No one knows a prescription drug’s side effects like the person taking it.
Make your voice heard.
RxISK is a free, independent drug safety website to help you weigh the benefits of any medication against its potential dangers.

Could it be my meds?
All drugs have side effects, but people often don’t link the effect they are experiencing to starting, stopping, or changing the dose of a drug. RxISK provides free access to information and tools to help you assess the connection between a drug and a side effect.
Explore our RxISK Zones
The RxISK Zones bring together a collection of content from our website about each type of side effect. They are aimed at giving you a good overview of the issue without having to search through lots of material.
PSSD Research Fund
Your donations are needed to fund scientific research into post-SSRI sexual dysfunction (PSSD) and other enduring sexual dysfunctions. The aim is to better understand the biology of these conditions and hopefully find treatments.
From The Blog…
Juggling Our Selves and Our Bodies
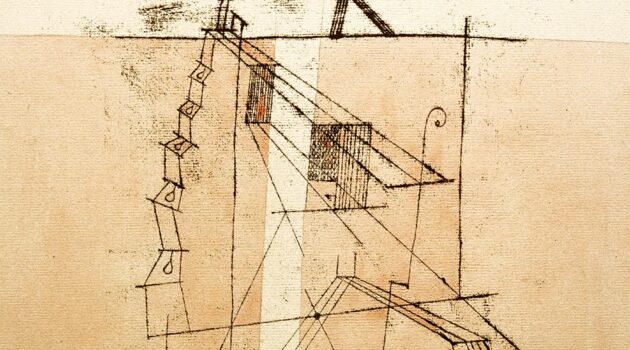
Illustration: Juggling Act © 2020 created by Niki McQueen Balance in Balancing Our Bodies and Our Selves and Juggling or coordinating Our Selves and Our Bodies are closely linked. Life is a Tightrope Walk and Paul Klee’s Tightrope Walker, emphatically concerned with his balance, features in Balancing. Once spotted Niki McQueen’s extraordinary Juggling Act had…
Ondine’s Curse: Can’t Live With or Without
Ondine’s Curse links closely to Balancing our Bodies and next week’s Juggling our Selves. Ondine’s Curse sounds like a Greek Myths. Its not. The story began taking shape a few hundred years ago and has been elaborated ever since – ending up featuring in The Little Mermaid and other stories. The standard version is that…
Balancing Our Bodies and Our Selves
Thanks to R who helped trigger this important RxISK post and some doctoring. . In July 2023, RxISK ran a post, written by R, giving an account of her Persistent Postural Perceptual Dizziness. PPPD was new to us. R had been in regular contact for a decade because of protracted withdrawal problems. Her problems came…