
This post follows on from Science, Kansas and Pancakes. Peter is doing a good job spreading word of this article on the Prozac studies in children and featuring other aspects of this extraordinary story. This post focusses in on one revelatory feature.
Reviewer #3:
A window with the view on the details of how drug approval proceeds. Very informative and helpful for understanding mechanisms of drugs (and vaccines!!) launching on the market.
This review – see Kansas and Pancakes – points readers to a section of the discussion called comment on context, which is laid out below. The results section and earlier Comment on the Results indicate that fluoxetine doesn’t work for children and is hazardous. The Comment on Context draws back the curtain on the Wizard of Washington. Almost no words in this are changed from the version in the article, but slides are added and some words to help readers for whom all this is new.
Comment on the Context
Given FDA recognition of a lack of efficacy on primary endpoints and significant safety hazards of fluoxetine, the results of this paper raise as many questions about the company, Eli Lilly, and the regulatory situation around 2002, as they do about fluoxetine use for minors. Aspects of the context in which these trials took place may shed a light on these company and regulatory issues.
Prior to the development of the SSRIs, there had been 15 randomised trials of tricyclic and related antidepressants in children and adolescents, all negative. An initial trial of fluoxetine was also negative. None of these were high quality trials. There was hope a well-done trial might demonstrate a benefit.
There was also, however, a clinical consensus and literature that children did not get endogenous depression. They might be miserable and unhappy, but this was situational and would respond to supportive interventions. Linked to this, there were almost no child psychiatrists with expertise in psychopharmacology.
A further feature of SSRIs at that time is that they were not effective in any age group for endogenous depression (melancholia). They had an anxiolytic, or serenic, action. The SSRIs became antidepressants in part to skirt around clinical concerns that any new anxiolytic would necessarily produce dependence as the benzodiazepines had. The RCTs done in children support the point that they are essentially anxiolytic rather than antidepressant.
Setting aside concerns about how companies did these trials, there appears to be better evidence for efficacy in conditions like obsessive-compulsive disorder (OCD) than for depression, although similar safety issues warrant concern about widespread use.
Erick Turner Act One
In 2008, in a now famous study, Erick Turner, who had previously worked in FDA, noted that of adult trials done as part of a licensing application for SSRIs and related antidepressants, 31% of trials viewed by FDA as negative were published as positive.
These positive publications are what make sertraline one of the antidepressants that guidelines recommend and that Andrea Cipriani has extolled to high heaven saying this is the drug people should be using – see Infection Super-Spreaders, Hengartner, Cipriani and Frontiers.
FDA made no comment about these publications, or about the later Emslie fluoxetine publications.
In 1997, the year X065 (the first Emslie Prozac in children study) completed, Congress offered half a year of patent extension to companies who submitted trials done in children to FDA. The studies did not need to be positive; the stated intention was to help establish the safety profile in children.
Two trials are needed for a license and Lilly immediately began study HCJE (a second Emslie study of Prozac in childhood depression). Both studies were submitted to FDA in support of a patent extension FDA supported this and also licensed claims that fluoxetine could be used to treat depressed children, as did other regulators that year.
Internally FDA had concluded that these two studies were negative.
After licensing the fluoxetine claim, based on studies negative on their primary endpoint, in October 2002 FDA issued an approvable letter for paroxetine in the treatment of children and adolescents who were depressed. The approvable letter is on Study329.org.

FDA’s letter agreed with GlaxoSmithKline (GSK) that all three trials they submitted (protocols 329, 377 and 701) in the application were negative as regards efficacy (letter available on study329.org). FDA also noted:
“Given the fact that negative trials are frequently seen, even for antidepressant drugs that we know are effective, we agree that it would not be useful to describe these negative trials in labelling.”
In the initial 2001 publication of study 329, a trial of paroxetine in depressed minors, GSK claimed paroxetine was safe and effective. The study (Study 329) was not dissimilar to the Emslie studies in terms of safety and efficacy. An internal GSK document from 1998, however, made it clear that the company knew the study demonstrated its drug to be ineffective but that it would be commercially unacceptable to publish this. This document also available on Study329.org states that
“the good bits of the study would be published”.
Based on this information, in 2004 New York State’s Attorney General lodged a fraud action against GSK. The settlement of this action made it possible to access data on study 329 and restore it in a manner that demonstrated paroxetine’s lack of efficacy and a trebling of suicidal events acts compared with the original publication.
This was published in 2015 in the BMJ – Restoring Study 329.

This information also triggered a crisis centered on the use of antidepressants for children. FDA convened a Psychopharmacologic Drugs Advisory Committee meeting in February 2004. It and other regulators published papers on 15 trials that had then been undertaken in depressed children and adolescents. The publications, including the Prozac ones, all claimed the drugs worked. The regulators at this hearing decided none of these trials (bar fluoxetine) demonstrated efficacy. There was a strong safety signal, a doubling of the risk of suicidal events, but FDA played for time and deferred action on this to a later hearing in September.
By then GSK had agreed to resolve their New York Fraud case and offered to make their clinical trial data public. This, and several reviews which returned figures for a doubling of the suicide event rate on antidepressants compared to placebo, led to an acceptance of the need for a Black Box Warning, in part because of the accepted lack of efficacy for antidepressants in this age group.
The licensing of paroxetine and other drugs was aborted but the approval of fluoxetine was not rolled back. All regulators, then and since, and medical authorities continue to claim that fluoxetine has been is effective in this age group.
To this day, the major media outlets like the BBC or medical journals like the NEJM are scared to say anthing other than this. See In the Name of the BMJ and In the Name of the BBC. Witnessing just how Lilly-Livered well-placed figures of the establishment can be has been astonishing. It is not as though the evidence they refuse to go near is hidden away – it lies there in the broad light of day.
The material available to us in respect of X065 (Emslie 1) and HCJE (Emslie 2) was less comprehensive than that for study 329 but our re-analysis makes clear there was no basis for claims for efficacy or safety. It can also be noted that Lilly and other companies have used fluoxetine as a comparator in subsequent antidepressant trials in this age group and that none of these studies demonstrated efficacy or safety for fluoxetine.
There has been one independent trial of fluoxetine, the National Institutes of Health’s Treatment of Adolescent Depression Study (TADS). This claimed efficacy and safety for fluoxetine, but efficacy was minimal and there were 34 suicidal events on fluoxetine compared with 3 on placebo. In none of 15 articles about this study between 2004 and 2010, written by senior figures linked to child psychiatry, published after the Black Box Warnings on these drugs, have these fluoxetine suicide data been addressed.
Duke University where the trial data was lodged has refused to hand over serious adverse event forms from the trial that might permit a restoration of this study – see link to Jon Jureidini video on Post-Truth and Science in Science Kansas and Pancakes.
Cardiac Events in Children
The dataset from X065 and HCJE on QT intervals also has a regulatory context. At the time these trials were being reviewed by FDA, Lilly had submitted a license application for R-fluoxetine, which was ultimately withdrawn in part because of QTc interval problems. QT interval problems are clearly an issue with fluoxetine in adults, as they are with all SSRIs (The Reverse Dodo Effect).
There were marked changes to cardiac QTc intervals in these Prozac pediatric trials. In response to FDA concerns, Lilly argued that the statistically significant increase in mean QTc found with the initial analysis was the product of random variability. FDA’s reviewer responded that, with a P-value of 0.009, the result was, by definition, unlikely to be produced by random variability.
Despite telling Lilly publicly they were crazy, and despite Lilly withdrawing R-fluoxetine because it causes cardiac problems, FDA did nothing else about this problem.
Erick Turner Act Two
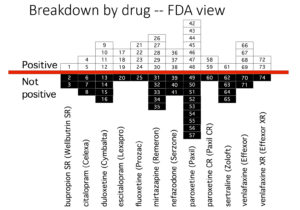
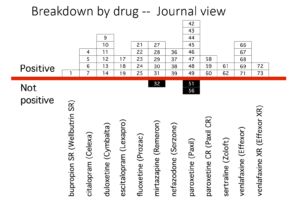
Erick Turner’s article is famous for two slides. The one further up the page shows how wonderfully well these drugs work. Who could resist prescribing them.
The second slide, here, shows what FDA saw and was perhaps even told by companies, as GSK told them in their children’s studies – the drugs don’t work in these trials, but agreed not to tell the world. These adult trials were done in th 1990s – before the trials in children.
Had FDA stated in the label for paroxetine given to children that study 329 was negative, it now appears they would have opened GSK up to a fraud action and a large settlement fine. This is exactly what happened when the internal company document came to light.
GSK and all companies are likely to have been aware of this when in discussions with FDA. It is not the job of drug regulators to police the medical literature but, if the medical literature limits a regulator’s freedom of movement, and forces them not to say things that we really should be told – both doctors prescribing and the rest of us taking – we have a nexus of issues that likely leaves everyone feeling uncomfortable.
If they play their cards right it looks like companies can almost force regulators to approve drugs on the back of negative trials.
When Emslie 1 and Emslie 2 were run, there was an expectation that many unhappy children could be helped with support. Support is still a good option. The approval of fluoxetine for depression in children and adolescents and the publication of many articles since, some of which have been shown to be ghost written, claiming efficacy for a number of SSRIs swept away the idea of relying on support and paved the way for a rapid increase in the use of antidepressants in children and teenagers.
Antidepressant drugs are now among the most commonly used drugs by adolescent females and among women of child-bearing years even though they double the rates of miscarriage, birth defects and behavioural abnormalities in their offspring. The figures for usage are as high in part because once started these drugs can be difficult to stop.
This is a public health emergency. FDA’s willingness to license Eli Lilly claims that fluoxetine is an antidepressant in paediatric populations was a key step in the evolution of this situation. The divide between what the academic literature on these drugs says and what the data when accessible shows is the greatest known divide in medicine. How this came about may need to be understood if we are to prevent comparable situations in future.
The Wizard of Washington
Several women, Shelley Jofre, working on BBC’s Panorama, and Cindy Hall, working at the Baum-Hedlund law firm, and Rose Firestein, working in the office of the Attorney General in New York, made the difference in this story. They rather than any academics, doctors or medical journals dug up the documents that brought a shocking scandal to light and derailed companies and regulators.
They ripped back the curtain that kept the Wizard of Washington concealed, revealing a pathetic old dude that we should not depend on to keep the system working or keep us safe.
The expectation many had at the time was that, given the scale of the scandal revealed, the system would be shamed into reform. Far from it. The Lancet, the Cochrane Collaboration and others we might think we could trust led the way in sweeping the scandal under the carpet.
Things are worse for children now, many of whom are on multiple psychotropic drugs. They are worse for the rest of us, an increasing number of whom are on drug cocktails that are shortening our lives and leading to evident falls in life expectancies in Western countries.
The regulatory apparatus has become an unlocked door against which companies or the Department of Defense can breeze through unimpeded. There are always some who think that this time the scandal is so shocking – as with Aduhelm or recent gene therapies – that things must now change. The system is inured to such scandals. Each shocking event makes it less likely there will be any change.
We’re off to see the Shrink,
The wonderful Chemical Shrink
We hear he is a whiz of a Shrink if ever a Shrink there was.
If ever, oh ever, a Shrink there was,
The Chemical Shrink is one becoz, becoz, becoz, becoz, becoz, becoz,
Becoz of the wonderful pills he gives
We’re off to see the Shrink,
The wonderful Chemical Shrink
Swallow the yellow-green pill,
Swallow the yellow-green pill,
Swallow, swallow, swallow, swallow, swallow the yellow-green pill.
Swallow the promise in the pill,
Swallow the capsule well or ill,
Swallow, swallow, swallow, swallow,
Swallow the yellow-green pill.
Apologies for the unscientific tone of this post. I must have caught a disorder from Peter Goetzsche.
To be continued next week.
People Acknowledgement
RxISK acknowledges that the experiences of those who have been harmed by medical treatments are the cornerstone on which it is built, and believes this should be the case for all of medicine.
See Black Robe, White Coat for more detail on this people acknowledgement

susanne says
1 of 1
Despite progress in data transparency, the FDA still keeps its data secret
Inbox
Maryanne Demasi, PhD from Maryanne Demasi, reports
7:25 AM (2 hours ago)
Open in app or online
Despite progress in data transparency, the FDA still keeps its data secret
MARYANNE DEMASI, PHD
NOV 10
History shows that hiding clinical trial data can be deadly.
Vioxx is a well-known example of how the US drug regulator withheld important information about the harms of the drug for over three years, before it was withdrawn from the market and tens of thousands of people died as a consequence.
Numerous initiatives have been launched over the past two decades to improve access to trial data after it became evident that what was reported in peer-reviewed journals was often cherry-picked and misleading.
Eminent scientists have succeeded in gaining access to trial data from the European and Canadian drug regulators, but a recent analysis published in the Journal of Law, Medicine, & Ethics, found that the US FDA still lags behind others when it comes to data transparency.
Europe ahead of the pack
Drug regulators have traditionally been the guardians of a treasure trove of trial data which they kept hidden from the public. But, over a decade ago, the efforts of Danish professor Peter Gøtzsche turned that on its head.
Prof Peter Gøtzsche, Institute For Scientific Freedom, Denmark
Gøtzsche and his PhD student were studying the effects of an anti-obesity drug and requested the trial data held by the European Medicines Agency (EMA).
“We already had good evidence that the efficacy and harms of drugs were incompletely reported in the medical journals, so by asking for the regulatory data for the anti-obesity pills, we were convinced it would get us closer to the truth”, said Gøtzsche.
At first, EMA denied their request, saying that it needed to protect commercially confidential information, but Gøtzsche was undeterred. He made a formal complaint to the European Ombudsman.
After an arduous 3-year process, the Ombudsman accused EMA of “maladministration” for refusing to share its data – it was a serious and embarrassing charge, so EMA had no choice but to capitulate.
In 2013, EMA announced that it would provide public access to regulatory data – which included study reports, protocols and the raw anonymised patient data in statistical programmes enabling anyone to independently scrutinise the data for all new drugs that it approved.
It was a bitter-sweet moment for Gøtzsche.
“I was satisfied with the outcome, but I also felt a bit betrayed. When EMA praised itself for being transparent, it conveniently omitted telling the public that it was basically forced to make the decision because of my efforts and that of the Ombudsman,” said Gøtzsche.
“I’ve been around a while to know that this is exactly how the drug industry operates. They cover up their failures while praising themselves for what others force them to do,” he added.
Millions of pages containing trial data have since been released. Interestingly though, this remarkable feat has gone largely unrecognised and the response from the research community has been rather tepid.
Gøtzsche suspects it’s because analysing regulatory documents is complex and requires experience to decipher regulatory data – skills that few researchers have.
“It is a huge job to do systematic reviews of clinical study reports held by drug regulators, but it is the difference between producing reliable reviews or merely “garbage-in, garbage-out” reviews,” said Gøtzsche.
Since then, Gøtzsche’s group showed this was the case for reviews of antidepressant drug trials.
When they compared data from medical journals to that from regulatory documents, they found major discrepancies such as underreporting of harms, including suicide attempts and aggressive behaviour.
Canadian regulator in the cross hairs
Following the landmark policy change in Europe, researchers believed it would help unlock regulatory documents elsewhere that were historically kept hidden from the public.
In 2016, Peter Doshi, professor at the University of Maryland and senior editor of The BMJ requested the release of unpublished clinical trial data relating to antivirals for the treatment of influenza (Tamiflu, Relenza) and three human papillomavirus vaccines from the Canadian drug regulator, Health Canada.
After some resistance, Health Canada agreed to allow Doshi access to the documents but imposed a confidentiality agreement that would prevent him from making his findings public.
When Doshi refused to sign the confidentially agreement, his request for access to the trial data was denied, so he filed a lawsuit in a federal court seeking a judicial review of the regulator’s decision.
Remarkably, in 2018, in the case of Peter Doshi v. Attorney General of Canada, a federal court judge ruled in favour of Doshi and in the public’s interest, ordering Health Canada to hand over the trial data for independent scrutiny.
It was hailed a “major victory” for transparency and after the win, Doshi told The BMJ, “For me this case has always been about something larger than my specific request. It is about the principle of transparency. If my case sets a precedent and Health Canada begins making clinical trial data available to others—promptly, and without imposing confidentiality agreements—that will be the real victory.”
Notably, the Canadian drug regulator has gone one step further than EMA by proactively releasing data for not only approved drug submissions, but also “unapproved, and withdrawn drug and biologic submissions…Class III and IV medical device applications.”
What about the US FDA?
The US FDA houses the largest known repository of clinical trial data in the world, but it doesn’t proactively share it.
In 2018, the FDA launched a new pilot program to proactively publish clinical study reports from the pivotal studies of nine recently approved drugs – but the agency put an end to that program in March 2020.
“It is just so typical of the FDA, which is very beholden to industry, and which some have dubbed the Foot Dragging Agency when it comes to the public interest,” said Gøtzsche.
Now, the only mechanism to ascertain regulatory data for FDA-approved drugs is to submit Freedom of Information Act (FOIA) requests, a lengthy process which often results in heavily redacted documents of limited value.
A study by US researchers, analysed the FDA’s willingness to release data, compared to other regulators, EMA and Health Canada.
They found that between 2016 and April 2021, EMA released data for 123 unique medical products, while Health Canada released data for 73 unique medical products between 2019 and April 2021.
In stark contrast, the FDA only proactively disclosed data supporting one single drug that was approved in 2018, clearly demonstrating that the agency has failed to keep pace with the European and Canadian regulatory bodies.
The problem of data secrecy within the FDA has been especially evident during the pandemic. Recently, I reported in The BMJ that the agency had failed to disclose covid-19 vaccine ‘safety signals’ derived from post-marketing data.
Also, the non-profit group, Public Health and Medical Professionals for Transparency had to sue the FDA for access to trial documents used as the basis for licensing Pfizer’s covid-19 mRNA vaccine. Initially, the agency wanted 75 years to release all the data but a Federal Court Judge rejected its request, ordering the release of the documents at a rate of 55,000 pages per month, taking approximately 8 months.
Given the widespread use of this important public health intervention, and the billions of dollars in public funds used to conduct vaccine research and development, these data should have been made publicly available immediately.
Data secrecy has undermined the health care system by subverting the allocation of scare resources and eroding public trust. The damage done to people’s confidence in vaccines, and medicines more broadly, will be felt for generations and likely to harm public health.
Donate Subscriptions
Share
Maryanne Demasi, reports is a reader-supported publication. To receive new posts and support my work, consider becoming a free or paid subscriber.
annie says
‘a morality play in SSRI-landia’
‘It’s the process that turned a limited and fickle drug like Prozac into a blockbuster by denying and hiding its foibles.’…
it’s not our drugs…
Emergence of Intense Suicidal Preoccupation During Fluoxetine Treatment
by Martin H. Teicher, M.D., Ph.D., Carol Glod, R.N., M.S.C.S., and Jonathan O. Cole, M.D.
American Journal of Psychiatry. 1990 147:207-210.
Six depressed patients free of recent serious suicidal ideation developed intense, violent suicidal preoccupation after 2-7 weeks of fluoxetine treatment. This state persisted for as little as 3 days to as long as 3 months after discontinuation of fluoxetine. None of these patients had ever experienced a similar state during treatment with any other psychotropic drug.
“Antidepressants occasionally promote suicidal actions in severely depressed-patients by enhancing drive and counteracting psychomotor retardation. However, standard antidepressants are not known to induce severe and persistent suicidal ideation in depressed patients free of such thoughts before treatment. We have recently observed several complex patients who appear to have had serious paradoxical responses to fluoxetine that were characterized by intense, violent suicidal thoughts…”
Posted on Wednesday 16 January 2013
https://1boringoldman.com/index.php/2013/01/16/its-not-our-drugs/
‘it created a firestorm’ – see full text
It’s not that different from multiple other situations in medicine with all kinds of medications. So what’s the big deal? Why did Lilly, knowing about Akathisia even before the drug was approved, try to keep it such a secret? Why code suicidality in studies as something else? Why attack doctors who brought it to attention in articles? Why trivialize “anecdotal reports”? By any rational logic, the drug reps should be handing out Teicher’s article on their sales calls. “Watch out for Akathisia!” “It’s a rare but important side effect!” But none of the manufacturers or the KOLs seem to ever do that. They mumble the warnings at the end of the tele-commercials, or in the scrolling small print on the printed ads. They finance increasingly bizarre articles and meta-analyses claiming a safety and an efficacy beyond the medications’ possibility to deliver. And they do it every time, at least in this modern era. But if these modern psychiatric drugs were presented with their actual efficacy and their side effects had been accurately characterized, they would never have been blockbusters. They would still be on the market and would still be of value, but they would be used much more carefully, primarily by people experienced in their use. There would have been many fewer doses of Prozac sold to fewer patients. Profits would have been only modest, court settlements rare. It’s not Prozac that’s a problem. It’s just a chemical. It was the entrepreneurial orgy that accompanied it.
In the comments section to a recent post, I’m asked the question, “SO on balance: HAVE ANTIDEPRESSANTS DONE MORE HARM THAN GOOD?” followed by a confrontation, “What is this rally against the biological psychiatry? You can dig up as much crap as you want. [actually you can do it in any field.] But unless you can address the above, all you’ve got going here is a morality play in SSRI-landia.” It deserves an answer. It implies that the point of this blog is an attack on Biological Psychiatry or the antidepressants. I think that question is a false dichotomy, or maybe addressed at the intent of some other blog. I’d rather answer this question, “Have the antidepressants done more harm than they would have if they’d been used carefully, based on the honest scientific evidence?” I’d say “You’re damned straight they have! By a mile!” And as for “all you’ve got going here is a morality play in SSRI-landia.” That’s pretty much exactly what I intend – morality. And if being put off by those Lilly emails from the dawn of time where people are lobbying for accurate reporting of Clinical Trial data, but then caving in, saying “Of course, at the end of the day, we will do what we are told to do” is my being a goodie two-shoes, I accept the designation gladly and wish I’d known to put them on sooner. I still practice general psychiatry in an out-patient clinic and use these medications in that practice every time I work – at least some of them. With the depressed, I mention and describe Akathisia to every SSRI/TCA naive patient I prescribe for, and give them a number to call if they have any problem [and they do sometimes – have any problem and call]. I try to paint a realistic picture of what is to be expected from the medication and stop the drug if there’s no response. When people discontinue SSRIs, I taper them slowly and warn about withdrawal symptoms [my track record treating withdrawal symptoms by adding another drug is somewhere around zero]. I never use the short-acting SSRIs because the withdrawal is often so difficult. I learned to do all those things by reading studies and writing this blog [because I didn’t know all the dangers before]. I expect by national standards, I’m a light-weight prescription writer. No apologies for that. And in the time available in a charity clinic, I have a go at finding out what’s going on in the patients’ lives that might be making them or keeping them sick, and help if I can.
And as for “You can dig up as much crap as you want”, psychiatry would be a hell of a lot better off right now if there weren’t so much crap to dig up. Looks like the Augean Stables to me. This isn’t a “rally against the biological psychiatry” – it’s a rally against the Eli Lilly and friends that joined in the fun and the lying and way over-promised, minimizing needed truths. And as for our current drugs, I see them kind of like I saw steroids, anti-metabolites, anti-hypertensives, etc. when I was an Internist. If you learn how to use them well and stay on the light side, you can help a lot of people, even cure a few, but the potential to do some major damage is always in the room, so you’ve got to be careful. It’s not our drugs that are the problem. It’s not even Akathisia itself. It’s the process that turned a limited and fickle drug like Prozac into a blockbuster by denying and hiding its foibles…
‘It’s just a chemical. It was the entrepreneurial orgy that accompanied it.’ …
susanne says
Lilly has lost loads of dollars in this exquisite scam – so did shareholders, some of them must have known about Lilly’s disgusting history before investing
Twitter Blue scam account may have cost Eli Lilly billions of dollars
Jon Silman Nov 11, 2022 6:39 pm
tumblr twitter verification
Credit: Tumblr
Pharmaceutical company Eli Lilly is the latest corporation to take a hit from Elon Musk‘s chaotic Twitter takeover. A fake account impersonating the company, with a blue checkmark purchased for $8, tweeted that the company was now giving away insulin for free.
The fake account posted the tweet around 1:30 p.m. on Nov. 10 and it stayed online for several hours. While it was up it garnered thousands of likes and retweets. By Friday morning, the account’s verification status was gone and it was set to private.
However, the damage had been done, with LLY shares plummeting 4.5%, according to Investor’s Business Daily. Other health stocks took a beating as well. Here’s a screenshot of the offending tweet:
The company was forced to respond to the situation from its actual account. “We apologize to those who have been served a misleading message from a fake Lilly account.”
Insulin is a drug that was publicly funded and given away by its creator Frederick Banting in 1923. He refused to put his name on the patent, claiming it was unethical to profit from a medicine that saved lives. The cost of the drug has tripled over the last ten years, reaching $450 by 2016.
It’s gotten so bad that people ration their doses to afford the costly drug. In a lengthy thread about the topic, The Jewish Vote co-founder Rafael Shimunov called out Eli Lilly for its greed.
“Eli Lilly stock isn’t just tanking because of a satirical Tweet on Musk’s hellscape, it’s tanking because millions of people are asking why we have to pay for insulin, when it was made to be free. That’s why other insulin stocks are falling too.”
Former presidential candidate and Vermont Senator Bernie Sanders also singled out Eli Lilly.
Someone else tweeted that “it cost some hero $8 to evaporate billions in Eli Lilly stock value. Elon accidentally created one of the most cost effective anti capitalist tools in history.”
Sure the company lost billions but it’s hard to feel bad when there are people who literally only take half their insulin doses just to afford the costly drug. As for Twitter, there are lawsuits coming. So many lawsuits.
Dr Pedro says
Is Dr Emslie by any chance related to ‘GE’ in this article from BMJ Best Practice?
https://bestpractice.bmj.com/topics/en-gb/785
“GE has received research funds from BioMarin, Eli Lilly, Forest Laboratories, GlaxoSmithKline, and Somerset; has served as a consultant for Biobehavioral Diagnostic Company, Bristol-Myers Squibb, Eli Lilly, Forest Laboratories, GlaxoSmithKline, INC Research Inc., Lundbeck, Pfizer Inc., Seaside Therapeutics, Shire Pharmaceuticals, Valeant, Validus Pharmaceuticals, and Wyeth Ayerst; and has been on the speaker’s bureau for Forest Laboratories.”
His association with Forest Laboratories is interesting as in September 2010, the company agreed to pay $313 million to resolve allegations of civil and criminal liability. One of the pharmaceutical-related charges was a misdemeanor charge of illegally promoting the Celexa and Lexapro for unapproved uses in treating pediatric depression.
Pogo says
In Jungian psychology, there is to my mind a parallel with this 1910 coming of age novel of Dorothy and our current time. Back then it was opium that many people used to ease their emotional pains and torments. This is alluded to in the novel when Dorothy and her companions are trying to find their way back to the yellow brick road to enlightenment. The wicked witch of the west conjures up a poppy field which causes Dorothy to abandon her quest (symbolized by falling asleep). Today, opium has been replaced by SSRI’s for those suffering disunity with the world ( or the State of Kansas) in which they find themselves.
I find it coincidently interesting too, from another angle, that the Wizard of Oz has been mentioned in this article.
Something that has been going through my mind since I first heard Jordan Peterson explain that the hierarchy of lobsters is dependant on serotonin. [1]
A lobster that loses a fight is a depressed crustacean. (I’m paraphrasing heavily for brevity). Yet, if you give him serotonin, he soon gets back into the fight for dominance over other lobsters.
So say Dorothy had been a boy (say Derrick) and his aunt had been a widow, leaving Derrick without a male role model who set behavioural boundaries. Would Derrick’s archaeopsyche ego (represented by the Cowardly Lion who also posses the ‘Activator Temperament’) sudden, not only find courage (and its companion ‘anger’) if he found himself in a field of St. John Wort (taking literary licence here, as this herb is a SSRI) instead of poppies? And what if the Tin Woodsman without a heart (representing Derrick’s neopsyche ego) had been staved of loving care. After all, he was rusty, stiff and so unable to use his axe to chop problems down into manageable size let alone do it with loving care. And what if the Scarecrow (representing Derricks exteropsyche ego) was not competently clueless but had memorised mainly violent solutions to life’s many complex problems without any compensatory influences from a father figure.
And what if Derrick did not have the benefit of guidance from Glinda (therapist) the good witch of the North?
Would the field of St. John Wort have caused Derrick to turn away from his path and try to relieve his angst by taking out as many of the Munchkins (representing the common people) as he could in the style of a modern day mass shooter?
[1] Lessons from Lobsters | Jordan Peterson
https://www.youtube.com/watch?v=5ZOkxuNbsXU
Bob Fiddaman says
Ah, Dr. Graham Emslie, the same guy who was reprimanded by the Texas Medical Board after it learned one of Emslie’s patients, an unnamed male, died by suicide whilst under his “care”.
Emslie prescribed stimulant and sedative medications to the patient for several years without appropriate evaluation, monitoring, and medical record-keeping. The patient went on to kill himself. Emslie claimed he knew the patient who was the brother of an acquaintance of Emslie’s daughter.
When asked for the 17-year medical records of the now-dead patient, Emslie told the Texas Medical Board the records could not be produced. Emslie claims he only kept “scant” records scribbled on a notepad. The board was also told that Emslie regularly prescribed benzodiazepines and amphetamines over the telephone without physically examining his patient. (remote prescribing)
More here – https://fiddaman.blogspot.com/2017/10/karma-catches-up-with-dr-graham-emslie.html#.Y25L3HbMJPZ
annie says
Let them eat Prozac…
Dr. John Virapen, Eli Lilly and Prozac
https://fiddaman.blogspot.com/2011/08/dr-john-virapen-eli-lilly-and-prozac.html#.Y257S8vP02w
Irish blogger Leonie Fennell has a fascinating post up regarding the SSRi Prozac [fluoxetine]
Her post focuses on a recently released video where Dr. John Virapen, who worked in the pharmaceutical industry for over 35 years, opens the seedy door to the psychiatric and pharmaceutical industry.
Leonie’s Blog
Prozac, Eli-Lilly and bribing the Swedish government.
https://leoniefennell.wordpress.com/2011/08/03/prozac-eli-lilly-and-bribing-the-swedish-government/
Let them eat Prozac
On the face of it, the investigation of possible hazards posed by SSRIs does not seem to have followed the conventional dynamics of science, where anomalies in the data are supposed to spur further investigation. In this case, debate has been closed down rather than opened up. Journals that might have been thought to be independent of pharmaceutical company influence have “managed” not to publish articles and the appropriate scientific forums have “managed” not to debate the issues.
Is this evidence of undue pharmaceutical company influence?
Is it evidence of the power of the current epidemiological paradigm in medicine that effectively only values one form of evidence – that stemming from Randomised Controlled Trials?
https://www.healyprozac.com/default.htm
Archive for Prozac (fluoxetine)
https://ssristories.org/category/drug/prozac-fluoxetine/
SSRI Stories…
JJ says
Those drugs turned my family member into a zombie. Had a college degree, good job and family. Now can’t work or function in any way. No one takes responsibility. Profit is all they care about.