
Illustration: Is There Life After Meds?, © 2014 created by Billiam James
Here is a Question put to the recent RxISK post on Sensory Neuropathy.
I’m a psychologist and I have made numerous attempts to discontinue antidepressants only to experience significant withdrawal symptoms. I’ve been on antidepressants for 25 years at this point and have made four unsuccessful attempts at tapering. I have come to learn that most doctors have absolutely no clue how to taper and have no understanding that withdrawal is a reality that can be severe and long lasting.
After making an attempt to taper off of Effexor last year, I once again began to experience withdrawal symptoms which were mostly sensory in nature. When I started to experience autonomic nervous system symptoms, I then took it upon myself to find a neurologist to do a skin biopsy to determine if I had small fiber sensory neuropathy. She also sought to rule out MS (which I didn’t suspect). Of course, my brain and spine MRIs were normal as was my EMG.
The skin biopsy was positive for small fiber neuropathy which was upsetting but did not come as a surprise. I have significant reduction in the small nerve fiber density. Symptoms, of course, were relieved by the reintroduction of the medication. As far as I know, at this point I don’t have PSSD.
My question is: Is it the process of withdrawal from the antidepressant that causes the small fiber neuropathy or is it from the effects of the drug itself on the body?
Tapering
Some years ago, groups in Holland and Britain began to champion the idea of hyperbolic tapering of antidepressants – serotonin reuptake inhibiting ones in particular. The idea was to reduce the binding of SSRIs to serotonin reuptake inhibiting sites in a steady way, in part because the way binding takes place means that there can be dramatic changes in how SSRIs interact with the site and these changes may not be the same at all steps in the process.
This idea caught on. World Tapering Day is set for early November. One of the things that tapering and tapering strips can be credited with is helping doctors and others accept the reality of antidepressant withdrawal. This is easier for doctors to accept if they think something positive can be done to help.
It now sounds almost sacriligious to say there can be problems with tapering. But many people find it impossible to taper even taking years. Having been sold the idea that tapering solves everything, they blame themselves.
Others find themselves left with enduring problems – a set of difficulties now termed protracted withdrawal. Again they blame themselves – did I taper too fast?
Symptoms on Stopping
Antidepressant, antipsychotic and benzodiazepine withdrawal were all described by 1961. At the time opioid, barbiturate, and stimulant abuse and withdrawal had emerged as serious problems and these drugs of abuse captured the terms addiction, dependence, and withdrawal.
The problems these drugs caused were viewed as far worse than anything prescription drugs like antidepressants, antipsychotics or benzodiazepines might cause.
Some of us can stop antipsychotics without too much problem. Some can stop some antipsychotics but not others. Some can stop but have significant and pretty immediate problems – referred to as supersensitivity problems that can include supersensitivity psychosis.
But some of us develop enduring problems on an antipsychotic. The original problem described around 1960 was neurological and ended up called tardive dyskinesia (TD). TD is usually viewed as a motor problem. It emerges while on the drug and can sometimes be ‘managed’ by increasing the dose or reinstating the drug if the problem appears on stopping treatment.
There is a physical dependence here, and an element of tolerance, but no addiction, or craving. After stopping treatment, TD can endure for years.
The term tardive originally meant that this problem did not appear immediately after starting treatment, but TD has shaded over into meaning a protracted or enduring dyskinesia after treatment is withdrawn. Six decades of research has brought us no closer to solving the mystery of TD.
We now recognise that there are tardive akathisia, tardive dysthymia and other tardive states that withdrawal from antipsychotics can cause.
I wrote three papers on antipsychotic withdrawal in 1998, 1999 and 1999. These were efforts to smuggle antidepressant withdrawal into the literature – even adopting the term discontinuation syndrome rather than withdrawal to smooth the path.
[The comments following Antidepressants and Premature Death on the topic of smuggling things into the literature are pertinent here. In this case, the smuggling was into a journal edited by Dave Nutt – who was not inclined to believe any psychotropic drugs, even benzodiazepines, caused problems].
These antipsychotic withdrawal articles were about the tardive dysthymia, akathisia and what today would be called protracted withdrawal. This is now much more widely recognized with several RxISK posts on the topic that have hundreds of comments. See Sally’s Story, Still Treading Water and The Invisible Doctor.
Eli Lilly were busy at the time telling doctors about antidepressant discontinuation syndromes. Their target was paroxetine, whose short half-life supposedly caused problems that the long half-life of fluoxetine (Prozac), with its built in tapering mechanism, didn’t cause. This was marketing not science.
SmithKline (SK) and later GlaxoSK fought back claiming paroxetine was not addictive or habit-forming or dependence causing and that it had no withdrawal syndrome linked to it. DSM and most doctors and professional bodies went along with this word-play. All GSK would concede is that there can be ‘Symptoms on Stopping’ – SoS.
Shelley Jofre tackled this word play in the Panorama paroxetine programs. As she put it, most of us don’t want to know if SSRIs are addictive like opioids. We want to know whether we can get off them or not. If we can’t stop when we want, most of us figure we are addicted or hooked.
The Colour of Life
Ask a doctor if TD is a withdrawal syndrome and they will say no. Push them on what it is and they may uncertainly say nerve damage.
Like TD, Post-SSRI Sexual Dysfunction (PSSD) can start on treatment but is most notable on stopping. Like TD, it can last for years after stopping. It too looks like damage – although neither are irreversible.
What kind of damage? PSSD looks like a sensory neuropathy, affecting small nerve fibres. Numbness is its central feature, or pain/irritability in the case of persistent genital arousal disorder (PGAD).
Drugs are among the most common causes of sensory neuropathy. Alcohol was the best known cause for most of the twentieth century but in recent decades cancer chemotherapies have emerged as a cause. We can tolerate saying chemotherapies can cause sensory neuropathy because cancer drugs are supposed to almost kill us.
Chemicals appear to be the commonest cause of sensory neuropathies. These include all sorts of environmental toxins, prescription drugs and the recent COVID vaccines – see React19.org.
Just as TD is now recognized as only one of the tardive syndromes linked to stopping antipsychotics, so also PSSD and PGAD are almost certainly only two of the enduring syndromes linked to stopping serotonin reuptake inhibitors. There are enduring akathisia and dysthymia states here also.
There are a set of conditions often termed dysautonomia, a name that concedes damage, although hopefully not irreversible. Dysautonomia is common while on or while withdrawing from antidepressants. These are likely mostly sensory problems.
We don’t think of things inside the body as sensory but the autonomic system is primarily sensory. Out internal organs sense just as much as our external senses do and the vigilance systems in our brains pay more heed to sensory inputs from within than to those from without.
There are cardiac problems as outlined last week – Antidepressants and Premature Death.
Many cases of apparent urinary or prostate infections are interstitial cystitis (IC) which antidepressants can cause – see Girl on a Hot Tin Roof.
There are also, as we reported on RxISK years ago and more recently, vision problems that can start on SSRIs, get worse on stopping or only appear after stopping and that can endure.
TD is usually thought of as a motor disorder, but even it may be a sensory problem, with the disturbance lying in the small fibres in our joints mediating proprioception. Parkinson’s disease, often thought of as a motor problem, also comes with painful sensory symptoms and the dopamine agonists used to treat it cause protracted withdrawal syndromes with many sensory features (Dopamine agonist Withdrawal Syndrome).
There is another piece to the jigsaw. Antidepressants are known to have analgesic effects – that are not opioid or anti-inflammatory effects. They share this action with anticonvulsants like carbamazepine, gabapentin and pregabalin and benzodiazepines, all of which are used in sensory neuropathies to relieve pain.
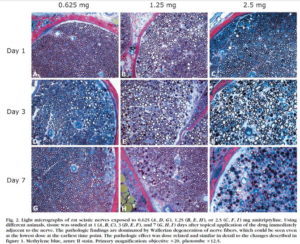
This gives rise to what we called Greg’s Dilemma in an earlier post. (There are a series of relevant posts featuring Greg – such as Riding a Bike Backwards). These drugs can alleviate painful symptoms occasionally – but at the price of potentially causing a more extensive problem over time. This image, featured in Greg’s Dilemma, shows amitriptyline working as a local anesthetic for rats causing nerve damage.
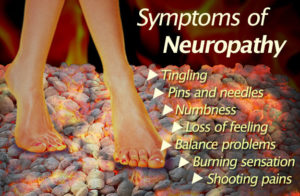
The image below is the traditional medical view of sensory neuropathy. Companies want to offer you a drug for this – Lyrica most recently – see Kicking Lyrica and The Day the Lyrics Lied. This is a very limited view of the sensory neuropathies which can involve every bit of the body and not just the feet and drastically impair our quality of life down to making us question if we are still ourselves.
A medical colleague got in touch some years ago, after finding to his surprise it was hard to get off the SSRI he was on. He eventually did and felt better but not good. Better because he could now do things the antidepressant inhibited. But he was still not right.
Several years later, rather abruptly, the colour came back into his life – he was normal again. This points both to the capacity of systems to regenerate but also to how profoundly disturbances to sensory systems can affect us.
Many with protracted withdrawal, or PSSD, report emotional numbing and depersonalization and see these as even worse than PSSD. These conditions are exactly what might be expected of a sensory neuropathy, which cuts off inputs to our brains that feed our emotions and givs colour to our lives.
Biopsy
The big problem for anyone with a sensory neuropathy is these conditions are subjective – like pain they hinge on the doctor believing you. Many doctors are slow to believe us particularly if we are complaining after being put on a pill for ‘nerves’.
RxISK attempted to solve this problem for PSSD by tracking down a machine to do Quantitative Sensory Threshold Testing. This led to a battle with a Parisian company – Impeto Medical and Device Wrecks – who have a QST machine and a genital adapter but they were unwilling to co-operate with us.
The alternative and possibly a better bet is a skin biopsy. As the recent Sensory Neuropathy post shows, several readers have got in touch reporting small fibre neuropathy (SFN) demonstrated on skin biopsy. This is very convincing.
But one comment on this post made clear that a reader with convincing PSSD had a negative skin biopsy.
There is however a paper that addresses just this point, describing a patient with genital neuropathy who had a negative ankle biopsy but positive thigh biopsy. This looks good but at present only ankles have a reference range – we can get results at the thigh and elsewhere and figure they look low but we don’t know what the usual figures are across all ages and sexes.
As the Sensory Neuropathy post made clear though, some discretion is needed when interpreting the results of any of these tests. Neurologists are not used to dealing with SFN in younger otherwise healthy people and a reference range taking in people on lots of drugs with loads of conditions may not be quite right for everyone.
The thigh result suggests a genital skin biopsy might be best of all but no one is likely to want to try one and there definitely is no reference range.
No-one either is going to want to go inside the body for a biopsy in the case of dysautomnia. In the case of vision though there is Corneal Confocal Microscopy which is safe and can be very convincing – but as another comment on the sensory neuropathy post shows that not all people with eye/vision symptoms will show up as having an SFN on CCM.
Still, it would be good to have a way to test genital areas directly and it remains important to get Impeto Medical to play ball. Hopefully readers can help with this.
If you approach a neurologist about a skin biopsy or try to get them to engage with a possible sensory neuropathy, they will suggest doing EMG conductance studies instead. These test motor systems and never show a problem with sensory nerves. It is mostly worse than pointless having one – as they will end up telling you there is nothing wrong with you.
Like most doctors, neurologists prefer the supposed objectivity of motor systems to the subjectivity of sensory systems.
Greg’s Dilemma
SFN can give clinical pictures consistent with both PSSD and protracted withdrawal from SSRIs, benzodiazepines, anticonvulsants and antipsychotics.
Those of us with protracted withdrawal, like the psychologist above, or PSSD, vision problems or dysautonomia should chase biopsies and SFN and report back to RxISK or somewhere that can compile evidence, which others can use when faced with neurologists reluctant to engage.
[Reports are better with names attached but reports to RxISK (of PSSD in particular) will remain anonymized. The offer to neurologists, however, may have to be that we can put them in touch with you, if you are willing, to confirm that you and others have a similar clinical picture to the patient they are now dealing with. The offer of contact rather than actual contact may be all that’s needed].
SFN is not caused by tapering too quickly, any more than TD is. Testing for SFN before tapering might save a lot of discomfort and heartache. It may be possible to relieve the distress caused by tapering in someone who has SFN by going back on the drug or raising the dose but going back on a drug that has caused nerve damage doesn’t sound like a great option – Greg’s Dilemma.
Those of us who can taper smoothly and are left with little or no residual symptoms are unlikely to have SFN, so not everyone with psychotropic drug dependence should think about a biopsy.
When SFN is present, we need to find ways to stimulate repair of these small fibres. The good news is that it looks like it can happen but may take months or years – as my colleague above found and several people with PSSD have found.
It is worth revisiting the posts on sleep disturbance during SSRI withdrawal. Disturbed sleep and insomnia (Sleep problems after stopping antidepressants, Insomnia The Royal Road to Pills and Nightmares, Forty Winks – and Brain Fog are all found with sensory neuropathies and probably add to the difficulty in making the diagnosis.
These seem Brain related rather than Sensory – and almost suggest keep taking your antidepressants and hypnotics. But they are more likely consequent on disturbed sensory input rather than brain damage.
It is worth paying heed to Bob Fiddaman’s account of managing protracted withdrawal, in the above posts, especially sleeplessness. He remained resolutely drug free and as active as possible. He was doing just the right things to get small nerve fibres to regrow, which they do, including avoiding making the problem worse by trying other drugs out.
Antidotes
Soon after the psychotropic era began, the anticholinergic group of drugs, an old and cheap group of drugs, were branded as the source of all problems. Just get rid of their anticholinergic effects and our new marvel drugs will work wondefully well and have no side-effects – like urinary retention, constipation, blurred vision or anything else.
In the case of the antidepressants anyway, these side effects are all serotonergic or catecholaminergic, not anticholinergic.
Anticholinergics in contrast are euphoriant and likely in a clinical trial of moderately severe depression would beat the SSRIs and be better tolerated.
Benztropine, the best known anticholinergic at one point, which used to be marketed by Merck as Cogentin was withdrawn from some markets some years ago. Not because of problems. It followed publication of a paper claiming that in low doses it promotes nerve fibre regrowth. Looks like Merck might have been thinking about tweaking it to get this effect, and claim they had a new pill for which they could charge a fortune.
See What to do about Sex for links to this article and input on pirenzepine, another anticholinergic drug that might help.
There were later many RxISK posts on Sodium and Transient Receptor Potential (TRP) channels exploring their roles in helping with withdrawal and PSSD.
We may have mistakenly in these posts slipped into viewing these herbs and other treatments as a way to ease withdrawal when they are better seen as possible treatments for enduring problems – once the provoking drug has been removed from the system. These posts are all worth revisting.
Another great comment on the Sensory Neuropathy post points to interactions between VEGF – vascular endothelial growth factor – and the serotonin system. There is a link in the comment reproduced here to a paper showing a role for VEGF in reversing diabetic neuropathy. What we do not know is why these rather old findings have not been picked up
These leads need readers to investigate them further. Having a goal such as finding mechanisms to stimulate small nerve fibre repair is invaluable for any search and holds out promise for a possible treatment.
The Side Effexor Withdrawal post over a year ago, and the three posts on venlafaxine and enduring withdrawal that followed it, introduced the idea of an antidote. A woman, Helen, who could not even begin to taper, found all of a sudden that introducing another antidepressant, dosulepin, freed her up to get off a crippling dose of Effexor rather easily.
The Invisible Doctor introduced the idea that some people who can stop all other drugs easily might have problems with quetiapine only – which can be eased by taking spironolactone. Since this report, I have been aware of others who have greater difficulties getting off quetiapine after being put on an ACE inhibitor for instance – which pulls in the opposite direction to spironolactone.
It is now clear the serotonin and Renin-Angiotensin systems (ACE) interact just as extensively as serotonin interacts with anything else – but we have been ignoring this for a long time.
Its worth noting, though, that spironolactone in its own right can cause akathisia. This is uncommon, more common in children, but definitely happens and is not something anyone mentions when talking akathisia.
These examples suggest that we should be looking for antidotes. Antidotes are not needed for uncomplicated psychotropic withdrawal, where tapering works well. They may help in clinical pictures involving neuropathies.
At this point though we do not know what happens if we then stop dosulepin or spironolactone. Do these antidotes facilitate withdrawal or remedy the neuropathy. Neuropathies can heal but not quickly.
Answer
This post opens with a question. The answer seems to be that many of these drugs can cause a sensory neuropathy. Withdrawal can reveal this but does not cause the problem.
Antidepressant dependence caused by people unable to stop for fear of feeling bad now amounts to a serious public health problem – with close to fifteen percent of most Western populations taking these drugs chronically. This is contributing to falls in life expectancy and a reduction of reproduction rates below the replacement level. It is also seriously impairing the quality of life of many of us.
Not everybody who takes SSRI or other psychotropic drugs gets a sensory neuropathy – although there are demonstrable sensory changes in everyone.
It seems likely that the higher the dose, the longer the duration of treatment, and perhaps concomittant use of psychotropic or other drugs, the greater the likelihood of a problem. Some drugs in each group may be worse than others or worse for some of us.
These are matters for which we do not have answers but answers could be assembled with a modest research effort.
Many of us can stop these drugs without much problem. Others can stop without too much apparent difficulty and tapering helps in these cases. Some however don’t feel good afterwards and opt to return to treatment. Some doctors looking at this might figure this person is psychologically addicted to their drugs but a contribution from milder neuropathic changes should be considered in these cases.
For some of us, but we do not know what proportion, many psychotropic drugs seem capable of causing marked problems and in many cases these may be linked to sensory neuropathies. Some have very focussed neuropathies – genitals or eyes. Others have much more extensive problems.
Oaklander Slides See the third comment below.
People Acknowledgement
RxISK acknowledges that the experiences of those who have been harmed by medical treatments are the cornerstone on which it is built, and believes this should be the case for all of medicine.
See Black Robe, White Coat for more detail on this people acknowledgement


Arjan says
As one symptom I struggled with was I couldn’t dream/imagine anything at all in the first few weeks post SSRI, aphantasia does not seem to be dependent on the sensory inputs. It’s now turned from just blank blackness into dimmed visions.
I wonder if the brain on its own can’t sense like before, it would reduce regeneration of sensors in the skin because it wouldn’t be needed like before…
“This points both to the capacity of systems to regenerate” — this might be due to neuroplasticity of the brain not regeneration of small fibers in skin
When I search for small fiber neuropathy symptoms, the most frequent one seems to be pain related, but somehow even though all my cognitive/memory symptoms, emotional numbness, skin numbness even in scalp…. is severe I haven’t had pain issue, Why is that?
Do all other small fiber neuropathy diseases cause pain?
Dr. David Healy says
Lack of dreaming is highly likely to be peripheral – a form of numbing. Nightmares are a common feature of withdrawal – see the sleepless posts – this is likely to stem from bodily irritability
D
Zach says
The blog is ok but two remarks:
1) Why is it so hard to not explain/list typical LONG KNOWN symptoms of autonomic neuropathy INCLUDING SEXUAL DYSFUNCTION (ed, lack of arousal, lack of lubrication etc). You talk of the “sensory” which is ok term.physiologically, BUT THE AVERAGE MEMBER OF THE SUFFERER COMMUNITY DOES NOT UNDERSTAND THE CONNECTION AND THINKS OF GENITAL SENSING ONLY. People are posting all the time about constipation, other gut motility related issues, lack of appetite, bladder issues, sweating changes, pulse changes etc. THEY ARE SCHOOLBOOK SYMPTOMS OF AUTONOMIC NEUROPATHY, BUT PEOPLE JUST CANT MAKE THE CONNECTION.
2) l think it is not smart to exclude /separate the brain side here, similar neurologic injur may take place parallel in certain brain tissues too.
Please consider these points in your communication. Validating more and more tested neuropathies would be the FIRST REAL MILESTONE EVER in the search for what these syndromes are related to. Research of the causes could then focus on the real issues. Public awareness could include this knowledge. Even law suits might become possible after enough validation. Now everything is dispersed and has no focus.
Dr. David Healy says
One of our Finnish contacts sent this comment
So it looks like you can get the same problems (“PSSD”, SFN, Akathisia and Tardive Dyskinesia) from both SSRIs and Covid Vaccines.
We are gathering data from the withdrawal group at the moment. Could we / RxISK take contact to Dr. Anne Louise Oaklander?
Dr. Anne Louise Oaklander from the Massachusettes General Hospital / Harvard happens to be the leading expert in this area. Somehow dysimmunity seems to be the case for most people with SFN as 80% of all cases responded to immunotherapy. She treats rapidly progressive “idiopathic” SFN cases with corticosteroids or IVIg with highly encouraging results. Giving severely sick people SSRI/SNRIs or epilepsy pills for just maskin pain doesn’t make sense.
Interestingly, the National Institutions of Health made a study where they treated Covid vaccine induced SFN patients with corticosteroids. The patients showed significant improvement in all neurologic symptoms after two weeks. Patients who showed persistent symptoms of dysautonomia and SFN for five to nine months were managed on intravenous immunoglobulin (IVIg) treatment. IVIg treatment was significant as symptoms improved in two weeks, resolving entirely for some and remaining as mildly residual in the others.
https://www.news-medical.net/news/20220519/NIH-team-documents-various-types-of-neuropathy-after-COVID-vaccination.aspx
Same results also here: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9128783/
Interestingly from 1988 we have a case report of complete recovery of tardive dyskinesia cases with corticosteroids: https://pubmed.ncbi.nlm.nih.gov/2898365/
Anne Louise Oaklander demonstrated success with IVIG / Corticosteroids after Covid induced SFN: https://www.reuters.com/business/healthcare-pharmaceuticals/nerve-damage-may-explain-some-cases-long-covid-us-study-2022-03-01/
We also have documented Akathisia cases after covid vaccine: https://pubmed.ncbi.nlm.nih.gov/34113842/
Tardive dyskinesia after Pfizer vaccine:
https://www.ehealthme.com/vs/pfizer-biontech-covid-vaccine/tardive-dyskinesia/
Dr. David Healy says
This all looks good but before getting carried away, RxISK has been in contact with Dr Oaklander starting 5 years ago to draw her attention to PSSD. She is a recognized expert on SFN and is especially interested in it in children. This is a relatively new state – perhaps it is linked to the greatly increased consumption of drugs in pregnancy in recent decades.
She is aware of PSSD but has shown no interest in it so far. We have tried high dose steroids in some people with PSSd with little or no benefits.
My hunch is that SFN linked to antidepressants or antipsychotics is not an auto-immune condition.
Our correspondent sent us some slides from Dr Oaklanders work – this are loaded up and appear as a link at the bottom of the post
DH
Ilana says
Here in the United States they don’t care to prescribe psychotropic drugs willy nilly and force them on citizens and visitors maybe bc they want their political way. It’s highly illegal for the cops to force or know someone’s health care at all but they are and Trump called to arrest them. They took the National Guard hostage using Healthcare and the Marines and other means. We’re living by Nazi propaganda here in Tennessee. It’s maddening bc it violates all our rights. They took over the courts and get their way with illegal conservatorships and all. It’s a nightmare! I wish the US could fight them but they are everywhere in my area.
Janie says
Sorry I don’t get what is the conclusion: is it worth to make a thigh biopsy or not in case no SFN detected from ankle?
Dr. David Healy says
J
I think it is worth it
David
Justin Oxley says
I began drinking a small 50ml measure of tart cherry juice which I diluted with water from 14th August. I was drinking this juice about a hour before I went to bed as it has been shown to raise levels of melatonin which helps with insomnia.
I was wondering if anyone with PSSD had tried drinking tart cherry juice for an extended period of time.
The paper below demonstrates drinking a daily measure of about 200ml of tart cherry juice produced measurable improvements in people with non diabetic peripheral neuropathies.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4566454/
Wouldn’t it be worth conducting a study similar to the one above on people with PSSD ?
Sarah Browne says
I would be very interested to know how these SFN cases are distributed across the two distinct types of PSSD: those who get symptoms of PSSD after a few doses of SSRI and don’t get better after cessation vs those who get PSSD only after stopping the drug.
I hope that when the cases of SFN are logged the subtype of PSSD is also recorded.
Dr. David Healy says
This is a good point. Once we have a number of cases, it needs to be explored.
DH
Serguei Novitchok says
Let me draw your attention to a recent study that may be relevant to your comment : Novel Autoantibodies in Idiopathic Small Fiber Neuropathy (https://doi.org/10.1002/ana.26268). The study established that patients with SFN have different characteristics depending on whether the cause of their SFN is known or not.
Sera from 58 SFN patients and 20 age- and gender-matched healthy controls (HCs) were screened against >1,600 immune-related antigens, using protein microarray. Patients with an unknown cause (idiopathic) of SFN had a different autoantibody profile than patients with an identified cause. According to the study, “Further subgroup analysis in the main cohort (iSFN and SFN by secondary causes (secondary SFN)) showed that MX1 was higher in iSFN compared to secondary SFN (HR = 1.61 vs 0.106, p = 0.009).”. I explained in a previous comment why I think an “autoantibody profile” study in patients with PSSD would be of great interest. It is likely that patients with PSSD having SFN have a distinct autoantibody profile from other patients with SFN without PSSD. Knowing the distinctive signature of autoantibodies in patients with PSSD may guide us to interesting leads, as specific autoantibodies may be characteristic of specific damage. As you suggest, different subgroups of patients with PSSD and SFN may also have distinct characteristics.
Sarah Browne says
Very interesting, thanks.
Sarah Browne says
It has also occurred to me that any differences in biomarkers in PSSD-related SFN and other forms of SFN are noted, not just to assist research on a pathogenesis and a cure. It is important to distinguish ‘types’ of SFN to get recognition of PSSD; SSRIs are taken by millions of people and therefore it is to be expected that some of them will get idiopathic SFN.
susanne says
This is just to alert people in England (I’m not sure if it applies to Ireland Scotland and Wales) that access to records is in a mess As historically some medics are still opposed and will be obstructive no doubt It matters as those who want a copy of their records will be given the runaround If considering giving a copy eg to Rxisk it would be an idea to get hold of them asap
This site is intended for health professionals only
(I consider we are all health ‘professionals’ and all info should be available to all)
Home News Technology GPs will have to re-redact records when patients move practice
GPs will have to re-redact records when patients move practice
Costanza Potter
17 October 2022
GP practices registering new patients will have to repeat the work already done by the previous practice of redacting their record, official guidance has said.
From next month, patients will automatically be able to read new entries – including free text, letters and documents – in their GP health record through the NHS App.
But official guidance has revealed that patients moving practice will lose access to any data from their previous surgery, meaning that if they request this again their new practice will have to repeat the process of redacting their records.
It comes as the BMA has called for the change to be delayed again and said it is having ‘urgent discussions’ around the new requirements, with guidance set to be issued within the coming days.
What does official guidance say?
NHS England FAQs set out that when patients register with a new practice, only new records from the date of registration will be visible, with any records previously accessed since 1 November lost.
The guidance, hosted on NHS England’s closed FutureNHS portal and seen by Pulse, said: ‘When a patient registers at a new GP practice from 1 November 2022, they will lose any access they had to historical information but will automatically get access to their future record in full from the date they join that practice.
‘If a patient would like to access their historical information (detailed coded record or full), they will have to request this and the new practice will need to review and redact if necessary.’
A local update from South West London integrated care board (ICB) to practices last week, also seen by Pulse, said that practices should alert the new practice to any entries they previously restricted since ‘online visibility settings’ are not part of a GP2GP transfer.
It said: ‘If a patient has some entries restricted online and leaves the practice, we recommend you have local procedures in place to contact the patient’s new practice regarding the matter.
‘Adding a patient warning would alert you to any restricted entries when, for example, you load the patient record.’
Meanwhile, the SWL ICB update also revealed that all EMIS GP practices will need to make changes to their clinical system to enable record access in order to avoid breaching their contract.
Practices that do not ‘enable their global system settings’ will be ‘unable to meet their current GMS contractual obligation to provide full record access upon written request and patients will not receive access to their prospective records in November 2022’, it said.
It remains unclear whether the requirement applies to practices using systems other than EMIS.
London GP partner and GP Survival chair Dr Nick Grundy warned that NHS Digital has not yet ‘put in place any of the tech required to make [prospective notes access] work’.
He added that it is ‘pure comms crap’ to say people have full access to their notes when they will ‘lose it all again’ as soon as they move practice.
He said: ‘The whole programme is yet another PR exercise by NHS England and one which characteristically seeks to solve a problem which doesn’t exist.’
‘Characteristically, again, NHSE piles a load of pointless bureaucracy on practices, without any functioning IT to support, so they can witter on about the “transformative” programmes they’re embarked on.
‘This is not transformative. It is a waste of everyone’s time.’
Speaking at the Best Practice conference in Birmingham this week, GPC England deputy chair and IT policy lead Dr David Wrigley said the BMA is ‘having urgent discussions’ and ‘will be issuing clear guidance early next week’.
Dr Wrigley told Pulse: ‘The BMA is concerned that the proposed model to centrally switch on default access to all GP patient records from 1 November will put patient data and safety at risk, as it is not consent driven.
‘There are also many concerns over the increased workload involved in this rollout and to expect GPs to review 61 million records is wholly unrealistic.’
He added: ‘We urge NHS England and Government ministers to reconsider the current go-live date and revisit the default access to records model for every patient in England.
‘We are, of course, keen to discuss possible solutions to design safe records access that works for everyone. As a consequence, we cannot support the project as it currently stands.’
The BMA previously said it had ‘not received satisfactory assurance that the citizen access to records programme scheduled to roll out on 1 November can go ahead’.
It said that the ‘necessary planning and resourcing required to launch the programme at this time cannot be put in place to enable a safe and successful rollout’.
At the same time, an NHS England blog this week committed to the rollout in ‘just under three weeks’ time’.
An NHS England spokesperson told Pulse: ‘Offering people access to their medical records is not only beneficial for patients in helping them manage their health, but it also helps practices manage demand better with people able to access information including their test results on their smartphones, where they feel comfortable to do so, instead of having to contact their practice.’
NHS England stressed that there has been extensive and ongoing engagement on records access and that it notified GP practices in July of the change coming on 1 November with all the detail they would need to prepare and comply by this date.
It added that it has worked closely with GP IT system suppliers to develop the required technical capability and that practices should contact their commissioner where they see challenges with providing the necessary safeguards.
As well as prospective records access, Pulse revealed earlier this week that NHS England plans to enable patients to request their historic GP records through the NHS App from next year.
Kosta says
There is a interesting paper from the 1970s from Richard a kunin where he succefully treated alot of his TD patients with manganese and niacin.
Phenodiazines apparently chelate manganese but idk about antidepressants and newer neuroleptics.
Also interesting that this hasnt penetrated to the mainstream sciences, maybe because he was a Orthomolecular doc and treated his schizos with nasty niacin with the horrible flush side effect. How dare he!!…