
Health Warning
I don’t know a great deal about the issues in this post. Experts by Experience and The Marketing of Anticholinergic Maleficence along with Psychotropic Drug Follies will give some feel for the background that makes it difficult to be certain about anything linked to the cholinergic drugs. The Past Present and Future article will need to be downloaded to take to your doctor for reasons outlined below.
The post does not make treatment recommendations. It covers a topic lots of readers are asking about aimed at enabling you to make your own judgement calls. It might help to consult others – not a doctor. Best people would be someone on or previously on an anticholinergic or cholinomimetic drug.
1980s Anticholinergics
During the 1980s and later, I and most doctors working in mental health gave out lots of anticholinergic drugs, usually by Brand Name. Knowing in this case means knowing by experience of seeing what the drugs did to people who took them and listening to what they said – as much if not more than knowing from academic articles.
The brands were well known without a drug rep handing out branded pens or stickies. This likely reflects the comfort most doctors and patients had with them. Being as comfortable as this with older, cheaper drugs typically leads to company vilification – usually through the medium of ‘responsible clinicians’, whose concerns get amplified in a way that never happens with concerns expressed on RxISK. From the late 80s onwards there was an increasing divergence between academic and patient level views about these drugs.
The main anticholinegic drugs in use in the UK and Ireland were:
- Benztropine, marketed as Cogentin,
- Biperiden, marketed as Akineton
- Procyclidine marketed as Kemadrin
- Orphenadrine, marketed as Disipal, Norflex
- Trihexyphenidyl, marketed as Artane, Trihexane, Tritane
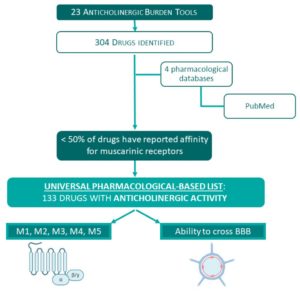
This short list contrasts with the 350 or so drug names you will find on Anticholinergic Burden IACB) Lists. ACB lists include drugs with a claimed anticholinergic action. Your ACB score is got by adding the anticholinergic scores of all drugs you are on. If its high you will have dementia in the morning.
But:
- Less than half the listed drugs have any effects on any cholinergic receptor.
- The effects of many are minimal compared to proper cholinergic antagonists.
- Many proper cholinergic antagonists do not cross the Blood Brain Barrier –
See this astonishing article – Lavrador et al..
The other 300+ drugs on these lists do cause dementia and other problems but its their serotonergic, dopaminergic and gabaergic actions which do this – especially when combined. Both pro and anti cholinergic system drugs are cognitive enhancing when used properly.
Withdrawal and the Cholinergic System
By 1961, imipramine and amitriptyline, the earliest antidepressants, had been reported to produce withdrawal reactions. Clinicians were warned that if these reactions cleared up on reinstating the antidepressant doctors and patients might be misled into thinking these drugs were more effective than they are.
These withdrawal reactions quickly got attributed to cholinergic rebound. The received wisdom was that cholinergic rebound like beta-blocker rebound would only last 48-72 hours, producing nausea, dizziness, disturbed sleep and nightmares. Strangely benztropine and procyclidine never seemed to cause withdrawal effects.
We now know that even for these older drugs withdrawal stems from their serotonergic effects and lasts much longer than 48-72 hours.
Antidepressant withdrawal is more complex than rebound. Rebound follows the expected dynamics of drugs separating from the receptors they are bound to – antidepressant withdrawal doesn’t. Rebound can be eased by tapering but can often be managed cold turkey and does not become protracted.
Tapering Antidepressants
Tapering antidepressants and using Tapering Strips to help people off antidepressants has been a great step forward. It encourages many people to think about stopping a treatment that was only designed to be short term but often lasts decades. Dee Mangin introduced the term Legacy Drugs or Legacy Prescribing to describe what happens.
Tapering as currently practiced has keyed into what we know about the dynamics of drugs separating from receptors. The implication that we understand the science of what we are doing reassures and motivates people.
But I don’t understand the science of tapering any more than the science of anticholinergic drugs. The images of antidepressants mapped onto hyperbolic curves used to promote Tapering are in part an artefact. The curves stem from the fact these drugs were brought on the market way way above doses that fully block serotonin transporters. It is no surprise then that titrating down from 60 mg of fluoxetine, paroxetine or citalopram to 20 mg should show little receptor binding changes. The surprise is that some people can find this terribly difficult and have even greater problems when they try to increase the dose back to previously comfortable levels. Something has become unstable.
There are other problems. Even tapering extraordinarily carefully and scientifically, some people can have terrible difficulties or find it impossible, while others can stop from huge doses relatively quickly – see Withdrawal Mysteries.
Then there is protracted withdrawal which seems to cover two different states. One is a terrible withdrawal taking a very long time as in Justin above. The other is a sense that yes I am off this drug, not suffering acutely, but a lot of things are not back to normal – with this latter state taking several years to respond – see The Colour of Life.
Antidepressant Toxicity
For me this suggests two interacting elements. One is likely to be a semi-classic withdrawal syndrome involving the drug separating from a binding site.
The other is antidepressant neurotoxicity. There is evidence serotonergic drugs choke or fry small peripheral nerve endings. This may underpin their ‘marginal’ benefits for pain. They degrade the fibres or sensory receptors mediating pain. Stopping the drug lifts the chokehold leading to a re-emergence of the symptoms of peripheral neuropathy that seem to map well onto the features of protracted withdrawal.
There are few articles on antidepressants and brain cell loss but there is evidence they cause osmotic demyelination – which is brain damaging. The radiological evidence that antipsychotics lead to brain cell loss is more compelling. Antipsychotics commonly, and SSRIs less commonly, trigger tardive dyskinesia – a state of significant neurological dysfunction if not damage.
If a neurotoxic effect is at least an element of what is happening, then part of the answer for some to getting off antidepressants and antipsychotics and perhaps anticonvulsants and benzodiazepines may lie in nerve regeneration. But until recently this seemed impossible.
Benztropine’s Disappearance
In 2012, a Scripps Research Institute multiple sclerosis (MS) study reported interesting findings – Scripps 1 and Scripps 2. Other studies have confirmed this.
The standard view on MS was that an inflammatory process attacks the myelin sheath protecting nerves. Treatments aimed at suppressing this inflammation included high cost monoclonal antibodies that also have a high cost in terms of lethal side effects.
When myelin breaks down, it is usually repaired by oligodendrocyte cells. But in MS these cells are missing in action.
Scripps screened old drugs to see could any stimulate oligodendrocytes and found benztropine did. Tested in animal models, benztropine reversed the defects found in MS. They said no other anticholinergic does this – so it was nothing to do with benztropine’s anticholinergic action.
It seemed the benefit could be found in very low doses which suggested some novel action beneath the headline cholinergic effects. This can happen – Josef Knoll’s famous studies on important catecholamine effects lying beneath the effects everyone thought were all there was is an astonishing story – See The Psychopharmacology of Life and Death.
Equally interesting was the fact Merck removed Cogentin from almost all markets – even China. They and others may have spotted a fortune to be made by finding out what the underlying effect was or a fortune to be lost if Monoclonal Antibody sales dropped.
Cogentin remained available in the US. A RxISK Nobel Prize post asked benztropine takers – but got no responses- whether:
- anyone with MS using benztropine figured it helps
- anyone on benztropine figured it makes a difference to something.
- anyone outside the US whose benztropine was stopped found they had developed new complaints since it was replaced by another anticholinergic.
- anyone could tell us the inside story on benztropine’s removal
- anyone knows how to get benztropine to people living outside the US
The Scripps effect was on big rather than small nerve fibres. Since then Paul Fernyhough in Manitoba and colleagues in San Diego, linked to WinSanTor have shown in animal models that another anticholinergic drug, Pirenzepine, regenerates small fibres in animals. WinSanTor have pushed forward with clinical trials and a press release, but not yet a publication, claiming pirenzepine produces benefits in diabetic neuropathy.
So it’s not just benztropine and not just big fibres but it is the anticholinergic action, acting perhaps to stimulate mitochondria back into action or to liberate VEGF which gets blood vessels to sprout with the nerves following the blood supply.
Another point to note is muscarinic – cholinergic drugs appear to modulate receptors rather than choke them like some of the SSRIs choke (bind with high affinity) to serotonin transporter sites.
There may be steps in between M-3 and nerve regeneration. Benztropine for instance has effects on ACE-2 receptors which SSRIs can knock out.
Another piece in the jigsaw was a 2017 report of a cholinergic dysautonomia in a 14 year old girl. This gave her an atonic bladder and bowel. She responded over a 5 month period to bethanecol – a cholinergic agonist for M-3 receptors. See Dysautonomia improving with Bethanecol. The authors figured that it was the agonist effect that helped but the time course of full recovery is consistent with nerve regeneration.
Muscarinic-3 (M-3) receptors are where the nerve regeneration action is these days. Muscarinic Acetylcholine Receptors
This poses a problem. Bethanecol is an agonist at M-3 receptors and benztropine and pirenzepine are antagonists – surely doing the opposite thing? In the bladder, the anticholinergic oxybutynin damps down bladder activity while bethanecol stimulates it – but both cases nerve regeneration may help.
The different actions may be resolvable. In the autonomic system, cholinergic fibres can be pre-ganglionic and post-ganglionic and each of these drugs may give opposite effects if acting pre or post-ganglionically. In addition in different cases – autonomic or peripheral neuropathies – they can appear to do the same thing – regenerate nerves.
The Treatment End
Benztropine and other anticholinergic drugs have been used for akathisia. They can help but are not particularly useful – red wine and beta blockers and P-5-P at least for some people are better. Bethanecol can be useful for sexual function but its not Viagra (although Viagra is not as good as the marketing hype suggests).
We are not looking for headline benefits like these – although the comments after the Visual Snow post raise the prospect of an immediate visual effect when eyes are dilated with an anticholinergic drug. Nerve regeneration is a slow process taking 3-6 months in all likelihood.
If the toxicity story is right, the nerves needing repair have been poisoned by high doses of drugs. They do not need toxic doses of a cholinergic or anticholinergic drug. At present, the research suggests the lower the dose the better. If a drug needs to be taken for months, you do not want it causing problems.
The first step is to aim at the lowest available dose and perhaps only one pill per day – think of it as more like a vitamin. This is a case of Less is More – along the lines of Knoll’s demonstrations in the psychopharmacology of life and death.
In all likelihood all anticholinergics will work but maybe not for all people. Pirenzepine is not absorbed into the body – it is used as a paste on skin or for gut problems.
Internet Pharmacy?
For people outside the US and Canada who want benztropine, the next issue is how to get it. I don’t know whether internet shopping is safe. Some readers likely do – please advise in a comment.
Off-Label
After that, whether in the US or elsewhere in the world, especially Europe or the UK, the next problem is persuading your doctor to give you an anticholinergic off-label.
Technically this is easy – doctors are free to prescribe off-label. The label of a drug is supposed to control the claims companies can make about their medicines. Labels are irrelevant when it comes to what doctors can prescribe. Many doctors, however, are bamboozled by this and will insist they cannot prescribe off label. See Healy and Nutt – Prescriptions, Licenses and Evidence. You can try bringing them this post and the H and N article. But it may be no good and you may need a friend to get it for you.
Even if your doctor is relaxed about labels, they might not want to prescribe an anticholinergic drug for you or a cholinomimetic (for Alzheimer’s) drug or something ancient they have never prescribed before like bethanecol.
This is especially the case for anticholinergics, which have been vilified possibly more than any other drug group in history. The most you can probably do is leave the articles and posts with them and let them think about it. We are talking about something in a very low dose that is probably less harmful than low dose aspirin, a drug that you would commit to stopping immediately if there is any hint it is not suiting you.
Who Does this apply to
Withdrawal
Anticholinergics are not an antidote to SSRI withdrawal effects. It seems most appropriate to try them if you have been able to get off an SSRI but have residual symptoms, especially if they are severe to the point of thinking you may need to go back on an SSRI.
Going back on a previously successful SSRI often doesn’t work. This is one more indication that something else is going on in these protracted states not typical of withdrawal.
If the problem symptoms seem more gut or cardiac (POTs) rather than altered sensations around the body especially in hands or feet, bethanecol may be worth considering rather than an anticholinergic. Gut and cardiac symptoms point to an autonomic neuropathy rather than a peripheral neuropathy. The case of the 14 year old girl noted above suggests bethanecol may work better for an autonomic neuropathy.
What to do for complex protracted withdrawals with both autonomic and peripheral neuropathy features? Who knows! Perhaps treat the more severe set of problems first.
A further group of people are those who have tapered down to a certain point and feel unable to taper further and contemplate going back up a dose. It is difficult to know how to advise them. Take a plunge and go cold turkey and then start an anticholinergic? Or stay on the same dose and start an anticholinergic and perhaps Taper later if it feels like it has become easier to do so?
I have had one lady in this state that benztropine added to her SSRI does not appear to have helped – but a lot of people in these states want rapid benefits or to ‘feel’ a benefit and saying you have to take this drug for weeks or months can sound like the advice many people got from their doctor about SSRIs in the first place that landed them with the problems they now have.
Enduring Sexual Dysfunction
A key group of people are those with PSSD, PFS and PRSD. I believe a decade back after the Nobel Prize post, I had a patient whose PSSD improved on benztropine, but I lost contact with him.
If there is going to be any benefit in any of these states it may take months to appear. It is also possible that bethanecol may be a better bet than an anticholinergic if PSSD and related conditions stem from an autonomic neuropathy rather than a peripheral neuropathy.
The 14 year old girl had an atonic bladder rather than an overactive one. If PSSD looks to you more like an atonic state rather than an overactive state, then bethanecol may be the way to go. On this line of thinking an anticholinergic drug may be more helpful for PGAD.
Having made these points there seems to be an interaction between cholinergic and ACE-systems that needs exploring further and Luisa Guerrini in Milan is the person to do this.
Bowel, Heart and Vision Problems
One of the most consulted and commented on RxISK posts was Bladder and Bowel Problems on Antidepressants Serotonin Reuptake inhibiting drugs cause Pelvic Floor Dyssynergia – a terrible problem that leads to suicide.
SSRIs also cause Gastroparesis – another debilitating condition, with to date no treatment. This looks like a good candidate for bethanecol.
Then there are the Vision Problems outlined in the Visual Snow post, some of which tantalisingly might even respond dramatically quickly to an anticholinergic drug – although it might need to be continued for some time to cement the response in place.
There are also cardiovascular problems like Postural Orthostatic Tachycardia Syndrome (POTs), triggered by SSRIs and Covid Vaccines it seems. Many people have had antibody tests done in conditions like these and antibodies to the M3 receptor turn up regularly.
Many of these Gut, Heart and Vision conditions just as with protracted withdrawal and PSSD often appear on stopping the drug and endure for ages afterwards pointing to a commonality.
The Nicotine System
Nicotine can be a wonderful cognitive enhancer and stress reliever – as good for some people as SSRIs for OCD or depression. Its not a treatment for PSSD. Sexually its more linked to erectile dysfunction although part of this may stem from smoking it.
There are a number of nicotinic antagonists like suxamethonium and rocuronium. Linked to this, a regular commenter on RxISK posts recently claimed ECT had cured his PSSD – and definitely cured his emotional numbing, anhedonia.
It is difficult to see how ECT would do this – it acts on motor rather than sensory systems. But to get ECT, you have to be put to sleep and also be given a muscle relaxant, either suxamethonium or rocuronium and these nicotine antagonists have profound depolarising effects across the body.
If anyone had relief from their PSSD, PFS, PGAD, gastroparesis or withdrawal states after an operation, let us know. You likely had suxamethonium or rocuronium when you were put to sleep.
The Vagus Nerve
The Vagus Nerve lies at the heart of all these issues. It is viewed in many quarters as the most important nerve in the body – key to making us sociable.
Anyone who wants to spin a good story about the matters dealt with in this post might being by acquainting themselves with Polyvagal Theory.
To Infinity and Beyond
After a recent lecture covering PSSD, a woman asked me if anyone had tried DMSO for nerve regeneration. I had never heard of Dimethyl Sulfoxide. Just shows how little I know so treat this with care – appearing it may at least stabilize nerve damage but of course as with all things available over the counter there is a lot of information out there telling us this is highly dangerous. It may well be for all I know.
Almost immediately after I heard first about DMSO, I heard about it again. It seems we may have discovered Life on a Faraway Planet . The evidence for this is the James Webb Telescope has found chemical evidence in the spectroscopy of this planet that looks highly like DMSO.



Katy says
I just have a layman question and I want to ask for opinions/ experiences. I was wondering if sacral nerve stimulation devises used for Bladder and bowel Incontinence, have ever been trialed or even considered to support PSSD? I am not a professional in the field, just a Protracted Withdrawal Sufferer. This may be a crazy question but I know people who would consider anything, to be relieved of this terrible condition.. I’m interested in your views and if anyone has experience of this in relation to PSSD.
Dr. David Healy says
K
This is a great idea. Would be great if some people tried it and reported back.
Darren in comments after some recent posts claimed he got a lot of benefit from lower back massage, which may be acting in a somewhat similar way to sacral nerve stimulation. It may also be that people who routinely use mechanical back massagers just don’t get PSSD. These option all need exploring and feedback
David
Bernard says
Doctor Healy,
Have you heard of the recent article on the mechanoreceptor PIEZO2?
https://www.science.org/doi/10.1126/science.adg0144
There are other less recent articles on the topic.
Do you think it might be involved in PSSD?
Dr. David Healy says
Yes – indeed – trying to get a copy of the article – have loads of the older articles
D
Isabella says
Hi Dr Healy
Have you had anyone report back to you on any kind of use of Benztropine since your last article on the drug?
I’m honestly surprised at the lack of discussion in all PSSD forums surrounding it, when we as a community seem to be so up for trying anything. And at the lack of suggestion of anything else by doctors.
Why have we not tried a drug that seems safe when some of us have tried Electro shock therapy, camel Urine, even reinstatement etc.
Dr. David Healy says
Isabella
Thanks for this note. Ideally we need feedback on several of these anticholinergics because benztropine is difficult to get in some countries. A second snag is having to take it for a connsiderable period of time without possibly noticing much if anything. Indeed if it is taken while still on an SSRI, this in its own right might cause problems.
Part of the reason to write the post is based on prior clinical experience with these drugs when they were being used far more sidely. I’ve known of people killing themselves taking SSRIs, SNRIs, noradrenaline reuptake inhibitors, dopamine antagonists, etc but cannot remember anyone coming to grief on a cholinergic drug.
I used to run healthy volunteer trials and some of the volunteers had significant problems on low doses of antipsychotics. SSRIs or noradrenaline reuptake inhibitors. I never did an anticholinergic HV trial but always figures that if I had to take something that seemed the best bet
D
Isabella says
There’s a large post on the PSSD forum website where a bunch of people tried Bethanecol as it’s more easily accessible.
2 were healed but one lied, and no one else saw any differences however, I don’t think they sustained the dose much beyond 2 weeks.
I think they ran into problems with more numbness which is a little scary, and a shame as it is much more easily available than Benztropine.
As desperate as I am to try anything, I don’t want to mess up my system anymore than I have with SSRI’s alone.
Thanks for your reply
Dr. David Healy says
I
Thanks for this – great to get some feedback even if third hand. It would be wonderful if someone can find out just what dose was used. Were the takers on anything else at the same time.
The ‘numb’ effect is interesting. Bethanecol in some ways should be like acetylcholine released from the Vagus nerve – it should be heart slowing and soothing Calming. Perhaps Bethanecol would be useful for PGAD.
In terms of the Vagus nerve – it has a branch to the Uterus and this apparently can mediate uterine orgasms when clitoral sensitivity and orgasms have been lost. If any woman with PSSD tries an anticholinergic it would be interesting to know whether she notices an inhibition of uterine sensitivity and vice versa with bethanecol.
Great if you can pass this message on and get anyone testing these drugs out to report any effects they notice.
D
Isabella says
Hi David
Thanks again for your reply.
It seems the dosage was mostly 4x25mg a day. A lady on the forum claimed she could feel her vagina again- so a resolution of genital numbness but still no pleasure. But everyone else declared no improvements although it seemed it was only trialled over 2-3 weeks.
The single case it ‘healed’ was unfortunately taking MDMA and mushrooms at the same time, which he claims however had no effect on his PSSD.
I’ve asked around in forums about this drug and a lot of old timers seem to respond with ‘it was the cure of the month a few years ago’. Disappointing of course but, perhaps trialling it for 1 month + would be beneficial.
…And I suppose it is not Benztropine, I’ve re mentioned this in the Reddit as it seemed to have gone a little unnoticed.
Hope to hear more leads from you soon
Thanks
Dr. David Healy says
I
Great thanks for this.
D
John says
Doctor
I’m looking up how to find benztropine. I reside in Belgium. I will see a neurologist for a skin biopsy for SFN. I will report the outcomes to RxISK. May I ask:
1. After 8 years of Venlafaxine XR + Sertraline combo on double the therapeutic doses, I have anxiety + restless leg + itching at night + hot flashes + erectile dysfunction + urinary retention + chronic bloating despite 22 months of abstinence now. Is it the right choice to check for SFN? Do I ask for thigh biopsy or ankle? Do I ask to test for autonomic neuropathy too?
2. AVAILABLE in Belgium: Bethanechol. Anticholinergics for bladder: Oxybutynin, Darifenacin, Fesoterodine, Propiverine, Solifenacin, Tolterodine. Additional anticholinergics: Biperiden, Procyclidine, Trihexyphenidyl, Atropine.
Which one above could sufferers in Europe and around the world try, if they had similar complaints as me? One pill per day of the lowest dose as well? For 6 months?
Respectfully
John
Dr. David Healy says
J
Sorry for slow reply – this went into spam. You have a very complex case.
I assume this is withdrawal rather than PSSD. If it is look at the RxISK post on Side Effexor Withdrawal – I think June 17 2020, where dosulepin appeared to help ths lady in question. This can be got in the UK – don’t know about the rest of Europe.
Bloating sounds like gastroparesis and might respond to bethanecol. Urinary retention can be because the sphincter to the bladder is closed which is what happens with catecholamine reuptake inhibitors – which venlafaxine can be in high doses. It also happens if your bladder has become atonic – in which case bethanecol is often used and helps.
I think in your case a test for autonomic neuropathy – dysautonomia is likely more important than a skin biopsy.
Re anticholinergics – biperiden, procyclidine and trihexyphenidyl are the ones to think about
David
An
Dr. David Healy says
Comment from LL
“The surprise is that some people can find this terribly difficult and have even greater problems when they try to increase the dose back to previously comfortable levels. Something has become unstable […] something else is going on in these protracted states not typical of withdrawal.”
Thank you so much for articulating this.
I’ve been reading around the topic for months but the way you stated this made my situation the clearest it has been to me this whole year.
48 hours after increasing my SSRI from 3mg back to 6mg back in March, I woke up grinding my teeth, horrific, gruesome images in my head and bizarre, phantom crawling sensations under my skin that have not abated. I had some of these already after withdrawing, but they became infinitely worse. Six months later I have not stabilised. I now understand why – I am not in WD, I am in “neurotoxicity”.
It has been impossible to describe the horror of this experience. Of knowing something is so very, very wrong, of trying to find one’s way back to the world other people live in, while living through sensory experiences so horrific and bizarre that they ought not exist. I feel things daily that are so foreign to the experience of being in a body, the same body I have occupied lifelong, I can scarcely believe they are happening, even as they happen.
“Something has become unstable” indeed.
I am fortunate that benztropine is available in Australia. My goal is to get to 2.5mg if I can manage the dizziness and balance issues, and then start it.
Dr. David Healy says
LL
This is an important comment that goes to the heart of a lot of difficulties. Your point at the end – my goal is to get to 2.5 mg if I can manage the dizziness and balance issues and then start it
A lot of articles on withdrawal categorise dizziness and balance issues as cardiovascular – such as a Choinard et al 2015 article that is widely cited on for instance Surviving Antidepressants. Im the case of the SSRIs, I think these are more likely to be neurological than cardiovascular. If it was imipramine or one of the tricyclics I might be more likely to think cardiovascular. In their clinical trials GSK coded these problems as neurological – although this might have been for devious reeasons.
Why mention this? Depending on which you or I thought it was, it might steer thinking toward bethanecol (cardiovascular) with neurological being more benztropine. But at the moment, this is not an expert view – its much more sticking a damp finger up in the air to assess which way the wind is blowing. It will take feedback from you and others for us to work out what the best options are.
The other key point is just what is going on when you increase a dose and things get worse. There are lots of views about this. One not mentioned in the post is that this comes from a direct drug effect being added into what was already going on – that the worsening is almost like adding in a new drug with the agitation and other problems that might arise from this. I think this perhaps leads Adele on Surviving Antidepressants to say akathisia is not part of withdrawal but is a direct drug effect, the answer to which is to reduce the dose again.
We don’t really know what is going on and there are no easy options. Having tapered slowly and successfully to a certain point it sounds completely counter-intuitive to go Cold Turkey from that point. But some people do and have a terrible short term experience but perhaps an easier one overall in the longer run. See the relatively recent RxISK post – Withdrawal Mysteries – Jon and Justin’s Journeys.
Keep us posted on what happens and your assessment of what it means
David
LL says
Hi Dr Healy
Posted a couple of comments on the weekend but they’ve not appeared.
In summary they said – thank you for your generous reply to my comment.
Have spent the last few weeks on both a Lexapro support group and a couple of Vestibular disorder support groups and spoken to upwards of 20 people who got vestibular damage from Lexapro and also Effexor. Most were diagnosed with Vestibular Migraine during withdrawal or some PPPD. Some were also diagnosed with POTS concurrently which suggests a cardiovascular component?
The improvement seems to take 3-5 years on average. Nobody has said they’re 100 percent back to normal but some people said they were 80 percent better after X years (too many years).
The most common treatment for VM is Amitriptyline/Nortriptyline which I am not keen on, or Effexor itself. Some success with CRP migraine treatments, vestibular suppressants, antihistamines etc.
A small Lexapro cut 3 weeks ago resulted in a doubling in the severity of my symtoms. Is there any use starting the Benztropine now on 4.5mg?
Also am requesting a SFN test at next Neuro appt. I have symtoms such as light sensitivity, burning sensation on face and neck, overactive bladder issues, IBS, low BP and low HR, heat and cold intolerance worsening over the last few years which I had not attributed to drug dysautonomia until now.
I am 33 and have been on SSRI’S since 15. I am also on low dose lithium for “ssri induced hypomania” and isotretinoin last year, spironolactone this year for lithium induced acne. The prescription cascade is the reason for my attempt at Lexapro withdrawl. (While on isotretinoin in 2022 I had total genital numbness). I imagine the isotretinoin did not help my vestibular case either.
Thank you again for your work.
Dr. David Healy says
L
This comment had gone into Spam.
You are asking a good question but I doubt if anyone knows the answer. Benztropine or a related drug will probably not help the lexapro withdrawal. Will lexapro inhibit benztropine from doing the good things it might be able to do – no-one knows.
Combinations of isotretinoin and lithium put you in territory few people have even been in and no doctor knows anything about. Will to give isotretinoin for lithium induced acne. If you opt to reduce lithium you need to take care – there can be withdrawal problems from this.
Throwing spironolactone into the mix suggests a very courageous doctor – again no-one can know what they are doing here – they definitely need to be guided by feedback from you as to what you think the combination is doing to you.
David
G says
If your most distressing symptoms are Depersonalization, Derealizatoin, mood and stress-intolerance issues, anxiety, fatigue etc would you go with Bethanechol or Benztropine?
With Bethanchol would take 25mg split over four doses a day?
Dr. David Healy says
G
If this is withdrawal rather than PSSD its not clear – see the response to the next comment from I. As you lay it out these symptoms sound more neurological than autonomic – maybe sound more neurological – so I would lean towared benztropine or any other anticholinergic.
Re dose of bethanecol – i think 25 mg spit in two AM and PM might be a place to start – see response to I above
D
Bernard says
Doctor Healy,
For someone who has less symptoms (mainly numbness), would you advice to take a lower dose of bethanecol? Could 5mg per day be useful or would it be definitely useless?
From one of your earlier posts (about benztropine) I understood that only very low doses would be advised.
Dr. David Healy says
Bernard
No one knows the answer to these questions. If we had the answers we wouldn’t have the problem. Bethanecol 25 mg per day is a low dose – it can be used is up to 200 mg per day or more.
It is also different to benztropine and other anticholinergics – it is an agonist – they are antagonists – and in both cases it may be something else about these drugs that helps – the right dose of which we just don’t know.
All we know is drugs acting on the cholinergic system do things tha might be useful – how we don’t really know
David
LJ says
I’m sorry to hijack this blog for a slightly tangential comment, but it does concern withdrawal and I’ve been meaning to ask these questions for ages–an article I’ve just seen has reminded me.
https://www.webmd.com/depression/features/antidepressants
It was published today, September 26, 2023, but it appears to be rehashing the same old harmful tropes. I have questions:
1. This article says it takes about a year to get the “full benefits” of antidepressants (or longer if the risk of relapse is high). What’s the evidence for taking antidepressants for at least a year and who funded it? What does “full benefits” mean?
2. Does suggesting people to take antidepressants for a year (I was told this by my GP, too) setting people up for more severe withdrawal? (As I understand it the longer you’re on them, the greater likelihood of bad withdrawal.)
3. If point 2 is true, could this be a factor in why symptoms have so often been falsely attributed to relapse (NB the article does not once mention the word ‘withdrawal’ so once again it’s still muddying the waters with talk of relapse), thus reinforcing the belief people need to take them forever (due to their faulty brains)?
In other words, since the manufacturers knew about withdrawal all along (e.g. see BBC Panorama’s ‘The Antidepressant Story’), does the insistence that people take antidepressants for at least a year stem from a deliberate strategy by pharma to induce more severe withdrawal symptoms in people, which can then be readily dismissed by doctors as relapse (that warrants long-term/lifelong antidepressants)?
Dr. David Healy says
The article is pure marketing copy. When the first guidelines for how long you take the drugs came out the recommendation was 3-6 months. This was for treating severe illness with drugs that seem now to be less likely to cause withdrawal.
Once you start SSRIs and SNRIs now you risk being on them for life. Some people can stop – others even if on them for no good reason and only for weeks may never be able to stop.
The deliberate strategy is to avoid the problems being noticed – if you are on for a year you and your doctor may forget to stop them
D
R says
For Benztropine, what would be considered a low dose that still has the positive nerve regenerative effect?
0.5mg? Or lower? Or would it require higher dosage?
Dr. David Healy says
Good question. No one knows the answer. If you are going to be taking them for 6 months you need to make sure you aren’t having side effects. I would start at 0.5mg and if no problems might go to 0.5 mg morning and evening. You need to keep an eye on whether it interferes with sleep or dreaming if taken later in the day.
D
R says
Thank you for your quick reply, appreciated!
If I can manage to get it (isn’t available in my country) I’ll certainly trial it.
Chad says
R
Message me. Let’s talk about Benztropine. I’m in the US
Mike says
Doctor Healy
Sorry if this question doesnt really relate to the topic of this blog post and I dont know if this is more of a question for a Urologist. But is there anything you would recommend from hard flaccid syndrome I have been dealing with sexual dysfunction from Vortioxetine which I stopped in December 2021 the closest thing to recovery is Hard Flaccid Syndrome. Because I live in Ireland its impossible to get a medication like Benztropine unless I am suffering with what the medication was intended for not Parkinson’s disease and not PSSD. But again is there anything you would recommend for HFS? I find that Viagra and Cialais dont work anymore for some reason.
Thanks
Dr. David Healy says
Living in Ireland has nothing to do with it. It is good medicine to treat a condition with a drug that will help – this was the reason to say you should download the Past Present and Future article and bring it and perhaps this post to your doctor.
Irish doctors are in general less likely to make the mistake of sticking rigidly to things like drug labels than doctors elsewhere.
Benztropine however may be hard to get in Ireland. See response to the comment from Stuart Shipko for more on this point
D
Stuart Shipko says
As you note, benztropine can be difficult to obtain. What about diphenhydramine, also known as Benadryl. It is over the counter and, in addition to being an antihistamine, it is known to have a fairly potent antimuscarinic effect as well. Might this be a useful agent in promoting regrowth of neurotoxic damage?
Dr. David Healy says
Stuart
Thanks for this. Diphenhydramiee does have anticholinergic properties but in addition to being an antihistamine is also the original monoamine reuptake inhibitor – inhibiting both norepinephrine and serotonin reuptake. The relatively weak serotonin reuptake inhibition added to an anticholinergic effect I think might make is very useful as a way to withdraw, Its got some similarities to dosulepin which has been very helpful for some people finding it diificult to stop an SSRI.
But this feels tricky for PSSD. Having said this – we – I you and all readers really should try to nail now how serotonin reupake inhibiting any of the anticholinergics are – benztropine, procyclidine, biperiden etc .
Finally on Reddit there is some talk at the moment of people being helped by cyproheptadine. This is an old antihistamine which is also quite anticholinergic. It is not an SRI – it blocks S2/3 receptors which increases appetite and a scandal in the early 1960s when it was promoted by Merck as the solution to starvation in the Third World. Its like mianserin and mirtazapine which some people with PSSD have claieed have helped them but this help has not generalized to everyone and is not consistent.
Rather than diphehydramine though cyproheptadine might have a better profile for what you are suggesting – at least for PSSD. We don’t however know if it causes its own withdrawal problems – mirtazapine certainly can
David
Genevieve says
What’s the difference between blocking serotonin (Cyprohepadine) and inhibiting it (SSRI)?
Would cyprohepadine cause discontinuation withdrawal issues?
Dr. David Healy says
G
The key word in discontinuation withdrawal issues is the word issues. For some people we are talking about neurotoxicity or damage (hopefully not irreparable). This can happen on mirtazapines which is very cyproheptadine like – there are loads of people on mirtazapine dependence forums who have problems that overlap with SSRI problems. These can start after a few days of treatment and may still be issues years later or they can creep up on you slowly so that you don’t know you have a problem until you have been on the drug for ages.
Cyproheptadine encouraged some people because there can be a temporary improvement but if its nerve repair you want – which it might help with – you have to stay on it for months likely without little sign of improvement. This is not a risk-free option.
D
Drummer says
When will we have a cure for this? How many more lives will be lost? Mine will be soon!
Allen says
Hello Dr. Healy:
I’ve been of Fluoxetine for over a year and half and I noticed partial genital numbness: front part has some sexual sensitivity while the back (previously the most sexual sensitive part) is now completely numbness. There’re also some people in PSSD reddit mentioning partial genital numbness, can any existing theory explain this? Also, 3 weeks ago I tried Omega3 and multivitamin, which causes me severe depression and complete sexual numbness till now. Does it implies any endoctrine problem? I took the supplement for only several days and it gives me a lot of side effect similar to antidepressant.
Daniele says
Sorry if I connect to your question, but I had and I have the exact same problem, partial anesthesia from Prozac and then Vortioxetine, the latter was prescribed to me to try to remedy the side effects (POST-SSRI) of Zoloft and Prozac, with worsening of the sexual-cognitive-emotional clinical picture.
I tried to supplement after 1 year from discontinuation, DHA and EPA derived from algae, everything worsened after constant intake of 1 week: brain fog, genital numbness, decreased libido. I wonder why, are the damaged mitochondria of the cells that accumulate these substances and cannot dispose of them or act in some way on serotonergic receptors?
Thank you
Jeremy says
Should we just get penile implants and go on with our lives? I don’t know what else to do at this point.
Alex says
Hello doctor i would like to try Benztropine, what dosage should i take ?
Dr. David Healy says
0.5 mg once a day
D
Felix says
Dear David,
Antidepressants neurotoxic effects would explain the nerv issues and therefore numbness of different parts of the body. It would also explain autonomous dysfunction and symptoms.
But it does not explain why many sufferers experience a progress in their symptoms, long time after they have taken the last dose of the SSRI/SNRI.
In my case, I did only get numb genitals about 6 months after I quit. Now, 4,5 years later my symtoms are way worse and way more then in the previous years. I was diagnoses with severe SFN lately and for me it looks like this is an autoimmune issue; I get windows when beeing sick and the overall tendency is that its getting worse with time.
Now I experinece full body numbness and I am very afraid of the future. I am looking into autoimmune treatments, as I got crazy high antibodies in my blood.
Felix
Dr. David Healy says
Felix
Thanks for these details. You may be right – there may be some interaction between neurotoxicity and an immune reaction. An explanation when we have one will have to explain a profile like yours which is different to the usual – but this difference to what is usual might contain important clues to the usual
David
Felix says
Hi David,
Thank you for the reply.
There seem to be more and more cases upcoming of people who are expericing windows while beeing sick. Or people are now starting to notice this relation.Plus there are many cases who got worse after having covid.
Also many of the confirmed SFN cases seem to have progression in their numbness all over ther bodys. There are now around 20 skin biobsy confirmed cases of SFN and every week there are more coming. I assume the few negative cases could just be false negative, as there is i high properbility of being so. Having this information, what are the next steps to find out what exactly attackes the nerves, A clinical setup is needed to campare these cases I guess.
Felix
Dr. David Healy says
Felix
Recently, there have been articles about PIEZO proteins – when something like this comes out I write to the authors to see if what they have reported can be applied to PSSD. This is something you and others could do as well. If you aren’t in the PSSD Network you should join – they are doing great things to raise awareness of the condition.
The problem is no pharma company is going to get involved in helping establish what is wrong and how to put it right – no government is going to accept there is anything wrong and fund research to put it right. We have to be more like AIDS activists in the 1980s and force the pace
D
John says
I don’t want to keep spamming Healy with questions, so if anyone here has an idea, please let me know. Given that David mentions damage to the nerve endings due to psychotropics, it would be on point to avoid alcohol and sugar in our diet, right? To really provide them with the ideal environment to recover as fast as possible…
I’ve removed literally every delicacy from my diet, and I hope that I’m doing it for a good reason.
John
Christoph Muth says
Dear Dr Healy IAM from Germany 🇩🇪
I need advice, I’m dying alive. I have been taking Cipralex for many years. It reduced to 8 mg stupidly combined with herbal stuff in the past. As a result, I had a sensitive nervous system. In between I had COVID and two vaccinations from BioNTech. I was careful for a long time and after an accident I was switched to Cymbalta 30 mg. It was reduced to 20 mg during the creeping phase. After a short time, a tingling sensation like pins and needles appeared. This has become more and more frequent. Feelings of numbness also occurred. I started tapering it off with the neurologist. The tingling sensation became stronger after each intake and cramps and twitches occurred. I was given Gaba and later Revotil in a mini dose. I’ve been on about the last 20 pellets (equivalent to 5 mg) for about 4 months now. I now have cramps even in the mouth area, sometimes my left leg drags and I have tremors. In the areas where it tingles for a long time, it becomes numb, as if I had Botox in it. I still feel everything. However, the sweating is almost completely gone, even during sporting activities. In the evening, sometimes your hands or entire body parts fall asleep. I take Cymbalta in the morning and the body calms down for a short time. Then, after five 6 hours, he starts to vibrate, intensify again and convulse. I have now been diagnosed with small fiber neuropathy. The following was tried: Acupuncture Tens I take the following: High dose, vitamin D. Plus K every week. Vitamin C. Magnesium I get a glutathione infusion from the doctor According to doctors, it seems to me that the drug is neurotoxic to the small fibers, although several neurologists have confirmed to me that they actually regenerate throughout life. I get TCM herbs from a doctor that calm the nervous system. and tolerated will stabilize the mood and calm me down. However, it has no effect on the tremor and vibration or tingling. I go towards the nursing case fully conscious, even though I don’t have MS, anything or a tumor. Another stroke, another neuropathy where you can no longer feel anything. We are currently considering the following options with doctors: Switch to mini-dose fluoxetine The remedy must be removed from the body, if it causes damage, switch to herbal products from Ayurveda or TCM in a specialized clinic in Germany. I don’t want to break. How else do you calm your nervous system? I do intermittent fasting and it was suggested to me to try a ketogenic diet.
What can else I do ?
Dr. David Healy says
C
You may be doing too much. Most of the things you do are more likely to create more damage. For the body to heal itself and nerve fibres to regenerate you need to give them time free from too many chemicals
D
Christoph Muth says
Ok thanks what can we do we change to Fluoxetin? I waited to see the neurologist. I’ve been on one dose for three almost four months and the numbness, abnormal sensations, cramps. Twitches have now almost reached the entire body. I drag my leg. Despite hours of exercise like after the stroke over three months after each time cramps get worse due to the short half life. The tremor gives no peace, the vibration that goes through the body. That’s why the question of going with fluxetin with the longer half-life alternative would be to have a doctor use the appropriate herbs for TCM as an alternative that would at least stabilize me psychologically. The mood is good, but my entire body is almost dead. Lack of strength in the fingers, cramps at night and we can’t calm down the nervous system, even a higher dose didn’t help. I even have tingling in the mouth area as an emergency plan, if I just twitch, it’s almost like putting as last option the neurologist under anesthesia for a long time until my nervous system has calmed down sometime in months. It’s so bad I’m going to be buried alive. Hence the attempts to calm the nervous system. Every day is a huge struggle and the physical deterioration is so extreme
Dr. David Healy says
C
No-one can answer this without knowing your case in detail. Great detail. Human’s aren’t automobiles which are all the same – we are different and what might be a good answer for me might not work or might even harm you.
Whether they help or harm, all pills are poisonous – they are chemicals – and the hope is to bring good out of the use of these poisons – you have exposed yourself to more than the average amount of poisons – perhaps because you trust in the poisoners – perhaps its time to trust them less and try tuning into what your body might be telling you
D
Christoph Muth says
We have Only wait for over three month Only taken Gaba or small dosage of Revotril against tremor good food so Sport walking then the body small Fiber destroyed every singlee day more my doctors have tested all At first I was still sweating. Within three months the sweating had decreased to almost 5%, even when I exercised I only felt delayed heat in certain areas and so on numbness
Christoph Muth says
Hello Dr Healy, In my case, the situation has gotten worse for a while. My neurologist, who also follows holistic approaches, had thought about it and so I’ve been sneaking fluoxetine with 2.5 mg for four weeks now because of its half-life, which I then had in combination with around 5.5 mg of cymbalta. The nervous system calmed down after a short time. Tremor was almost no longer there and I’m an idiot. jumped to 2.5 mg with cymbalta. That was about three weeks ago now. The dose has been adjusted again. The nervous system doesn’t calm down and I still have fluoxetine in my blood. Tremor cramps at night not a quiet second tingling and neurological symptoms all over the body I hope the body calms down again. Because of the tremor, I’m taking small doses of revotril and herbal things in addition to infusion I’m really having trouble getting through the day. Although the mood is okay, it’s mainly the physical stories like pain, balance problems, dizziness, what’s the best thing I can do to avoid breaking down. In the long term, the goal is of course to taper off the cymbalta. First of all, stay on the dose and calm down the body. I do relaxation exercise movement now again I have head pressure pulling vibration in the face.
mike says
Dear Dr. Healy,
Im no native speaker and Im just a PSSD sufferer, no professional.
I maybe have something important to add in terms of pirenzipine.
5 Years ago (I’m 32 now), I was diagnosed with small fiber neuropathy/peripheral neuropathy,
probably because I’m suffering from Lupus.
I was researching the internet and found out about pirenzipine.
My uncle is a GP so he prescribed me the relatively safe drug.
I was taking 50mg for 3 month and then went back to the neurologist to perform a test.
When he saw my nerve speed test results he was completely shocked.
He said he has never seen anything like this: He admitted nerves recover but they recover very slowly.
I opened up to him and told him I was trying pirenzipine and he couldn’t believe it.
I never went back there and my nerves have been fine since.
…. So, pirenzipine doesn’t has to be taken topically, it can be swallowed.
I will try another round soon, to see if it helps.
Dr. David Healy says
M
Thanks for this feedback. Can you add it to a few more of the PSSD and anticholinergic posts – will approve all of the additions. Important people get to hear about this. Where did you get your pirenzepine from – if you can add this also it would help.
Someone else got it from Japan I think but then it stopped. They tried contacting WinSanTor but got no response.
David
Mike says
Hey David,
as I wrote:
My uncle is a GP so he prescribed me the pirenzepine (I’m located in Germany).
I’m willing to start a new off-label experiment (my uncle will prescribe anything safe), but I’m wondering which route to take: pirenzepine or Benzatropin.
Maybe you could help me decide and I will report my findings?
Some other symptoms:
– Since the onset of ED I also had strong sweating (although I’m super fit)
– My Urologist did a doppler test. he said my genitals look completely normal, but there was no errection although he gave me an injection. He said: the blood flow is normal but it seems as if the muscle tonus is too high. He said he has never seen anything like this and wanted me to perform autogenic training or other relaxation methods.
When my tonus is to high and that’s the reason for my ED (caused by Cymbalta), should I take the pirenzepine or Benzatropin route?
Dr. David Healy says
Mike
If pirenzepine has worked for you before, then I think it is the one to try again. Both may work in general but you have evidence pirenzepine works for you.
Where are you based in Germany and can you find out from your uncle how easy it is to get pirenzepine where you are and what dose to use
David
mike says
Hey David,
you may give my contact to Sandra, if she is interested.
Im from around Düsseldorf, my uncle is located in Berlin.
Pirenzepine is super easy to get; you just need an off label prescription. You can get it in every pharmacy.
But: as far as I understood your blog post. It depends on whether PSSD causes peripheral or autonomic nerve damage right? Pirenzepine regrows peripheral nerves as far as I understand. But the muscle tonus of the penile tissue is more an autonomic phenomenon right? …
Dr. David Healy says
Mike
Thanks for this – will link you up with Sandra.
Thanks for the information about peripheral and autonomic nerves – as I tell everyone the whole time, I learn everything I know from people who get in touch with details like this. Having said that, pirenzepines main use was to treat ulcers and the benefit it offered there looks like an autonomic nerve benefit rather than peripheral nerve.
We need to get a few people to try it
David
mike says
Last Thing David,
another interesting route just for the ED part.
As I stated my urologist noticed a high muscle tonus in the penile tissue which prevented the errection. He speculated a botox injection may help.
It won’t be a causal cure but it might help with ED symptoms.
Dr. David Healy says
These options need exploring
D
Tim says
This message from Mike has left me wondering. Did he get relief from his PSSD symptoms, or did his neuropathy improve just by the numbers?
Dr. David Healy says
Good question – have emailed him
D
G says
Discovered another, maybe more freely prescribed anticholinergic today: Scopolamine. It’s for travel sickness and comes in patches too. What are your thoughts?
Some people however say it causes them withdrawal.
Can’t use it myself and don’t know if it’ll work or cause more damage
Dr. David Healy says
Scopolamine is anticholinergic but quite different to benztropine. Its used for nausea and vomiting and causes cognitive problems and is generally viewed as risky.
Anna says
What do you think about treating the vagus nerve with so called transcutaneous vagus nerve stimulation? Could this help e.g. in antidepressant withdrawal?
Dr. David Healy says
This has come up before. I would need to look into it again to work out what I think but I think I thought it was unlikely to help. When we don’t know what the problem is, what’s going on to give rise to the problem. everything is s shot the dark
D
Carolina says
Hello David,
My name is Carolina and I’m based in Portugal. I made several bad decisions, and quit cold turkey 30mg Mirtazapine in December and went into a roller coster of withdrawl symptoms months after, that I didnt recognize.
In march a psychiastrist reinstalled 30mg of mirtazapine and I had a reaction exactly like Guillain Barre. I had a feeling of blood “ooshing super fast” as if something was entering my blood stream. Tingling, from toes to head, tongue numbness, incontinence, could barely walk, tongue spasms, visual problems, but the psychiastrist said it was psycosommatic. I had a horrible headache, i described it to my mom as if something was dying inside of my brain. It was a very weird sensation. I continued to take it, and continued to have the symptoms for 8 weeks, then discovered this was a toxic reaction and did a fast taper on mirtazapine 15mg. And stopped taking it.
While on the meds this second time and after leaving it 4 weeks ago I have abdominal bloating, burping all the time, my maximum blood pressure is 80 (sever hypotension) burning/itching/twitching that is turning into total body numbness with muscle weakness. My muscle is jelly like. Swollen lymph nodes. Super super dry skin and massive increase in facial hair (probably due to cortisol) and I have very clearly some cognitive impairment.
I will try to get assessed for polyneuropathy, as I have seen some people in reddit get IVIG and get better with the neuropathy part. But what hope can I get for the autonomic part? Will immunosupressors work for that department, or maybe anticollinergic?
Thank you so much for your time David and for all you do
Dr. David Healy says
Carolina
Your situation sounds grim. It is almost certainly not psychosomatic. This is a toxic reaction. It is quite possible all tests will come back normal which will make it difficult for you to maintain your claim that this is a toxic reaction and not psychosomatic – but even more important to maintain your position.
Several other people reporting to me/RxISK have had reactions to mirtazapine that are like this.
Readers of these posts and comments will know that I tend to suggest taking an anticholinergic. I don’t know that it will help you but I have no idea what will help you and the options are to do absolutely nothing to avoid making things worse or try something like this which is pretty safe.
As regards anticholinergic drugs, my suggestion at the moment is Oxybutynin possibly starting in a 5 mg dose in extended release form taken once a day in the morning. This is a low dose but you might at the moment be very sensitive to the effects and might need a lower dose.
Why Oxybutynin? It has some evidence that it can reverse peripheral neuropathy and is also thought to be anti-spasmodic.
It should be easy to get from a doctor – especially if you mention you are having bladder irritability problems.
If you can get it and try it please let us know if you are able to tolerate it early in taking. You may need to take it for several months to get the kind of benefit it might be able to produce – you should not continue with it if it is causing you problems. While it is generally safe to take, it doesn’t suit all people.
David
Carolina Varela says
Im having a problem which is Im also developing now tongue spasms and I feel like my tongue is bigger and I can only stay with my mouth open. Very bad swallowing as well. Im guessing this is like an extrapiramidal effect on top of the toxic effect I already had.
My doctor wants to change my medication for Trazodone. At this point I’m at a loss due to family and medical pressure.
Would the anti collinergic help the expiramidal effect of what I feel like is dystonia
Dr. David Healy says
C
Its very hard to tell without seeing you and even harder to know what might help without being able to follow you up when you try things. I still think based on what you’ve written that Oxybutynin is one of the better options. Try the lowest dose you can and don’t continue if it makes you feel worse
D
Carolina says
You think reinstating mirtazapine 15mg to stop extrapiramidal effects would make any sense?
Im not even worried about the nerve part at this point. I just dont want to have movement disorder + neuropathy
Sorry to disturb you so much Doctor!
Dr. David Healy says
No one knows the answer to this. You are better placed than almost anyone to decide. My instinct (but its instinct not expertise) is not to reinstate but if I was in your shoes my instinct might be different
D
Tim says
Hello David,
I would think that a prescription for oxybutynin can be arranged in my home country. What specifically makes this drug a more effective candidate in treating PSSD than benztropine in general? Are there any reports of patients who have tried oxybutynin and benefited from it?
I have been diagnosed with SFN according to international reference values and abnormal cytokine levels, so strong indications of an autoimmune disease.
Dr. David Healy says
It may not be better than benztropine – its more a case of availability. Initially it looked like there was a special magic to benztropine – it did something other anticholinergic drugs dont do but oxybutynin and pirenzepine seem to do it too – so the magic seems to be anticholinergic rather than some mysterious other factor
D
Carolina says
Hello Dr Healy,
So we went with reinstating mirtazapine at 7,5mg. I regained sensation to touch, and the numbness improved! But some other side effects muscular wise and movement wise are just unbearable. I still have some involuntary movements and nerve pain, the worst party of it all is really the hypotonia and hypotension. Now that I am tapering, and I am at 3,75mg the numbness is returning. It starts by feeling a burning sensation and after a couple of days the skin becomes more and more numb. But mine is generalized. It starts mostly on my hands and face. Genital área is the last to be affected.
Dr. David Healy says
Carolina
You are having a terrible experience. I know other people with similar effects on mirtazapine and it is difficult to know what to suggest. Please keep an eye on other posts on RxISK – the vision posts in the last week or two and other posts to come over the next week or two. and above all if you try anything and it helps or makes things worse let me know.
I hope to be lecturing about withdrawal problems on Mad in America in a few weeks time and might have some other options to mention by then.
David
Carolina Varela says
Thank you so much Dr Healy.
What I found bizarre and didn’t expect to improve was the numbness going away with reintroducing the drug, that is why I wanted to tell you. As I am tapering, I am getting more numb again, but for me it is not an option to stay on the drug so I will just have to deal with what comes afterwards anyways in regards to the movement part.
Thank you so much for all you do!
Dr. David Healy says
Carolina
Its not bizarre – its just not what the books say. The books say that raised blood glucose leads to peripheral neuropathy and pain and we have to get your sugar levels down – but there are loads of people with no pain or neuropathy but very high glucose levels who start developing neuropathy and pain when their blood sugars are brought down.
I think you said you were about 30 years old – do you have any variation in your symptoms depending on the part of the menstrual cycle you are in?
David